|
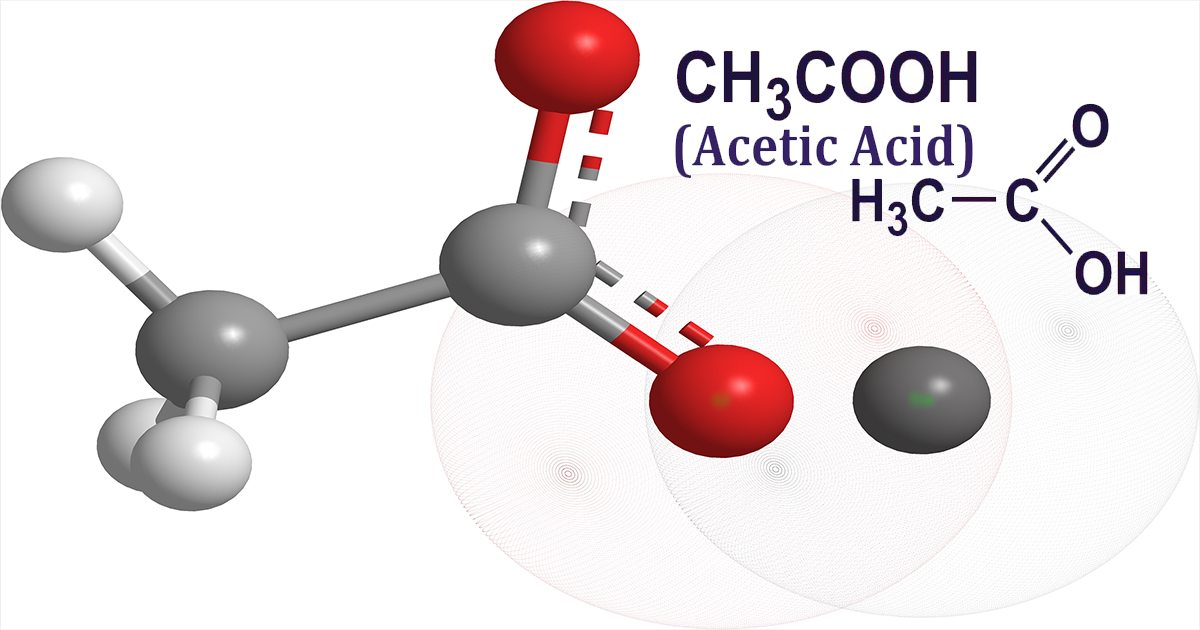
Like ethanol and water, liquid acetic Acid is a hydrophilic (polar) protic solvent. It dissolves not only polar compounds like inorganic salts and sugars, but also non-polar compounds like oils and polar solutes, thanks to its relative static permittivity (dielectric constant) of 6.2. It is miscible with both polar and nonpolar solvents, including water, chloroform, and hexane. Acetic Acid is not miscible in all compositions with higher alkanes (starting with octane), and its solubility in alkanes decreases with longer n-alkanes. Acetic Acid's solvent and miscibility properties make it a useful industrial chemical, such as a solvent in the production of dimethyl terephthalate. Read More- https://coherentmarketinsightsus.blogspot.com/2022/09/industrially-acetic-acid-has-been.html
0 Comments
Leave a Reply. |
Categories
All
|
 RSS Feed
RSS Feed
