Due To Its Low Sweetness, Polydextrose Is Ideal For Combining With High-Intensity Sweeteners17/4/2023 Polydextrose is a soluble fiber commonly used as a low-calorie bulking agent in food products. It is a synthetic polymer that is derived from glucose, sorbitol, and citric acid, and it is approved by the US Food and Drug Administration (FDA) for use as a food additive. Polydextrose has a range of functional properties that make it useful in a variety of food products. In this article, we will explore the various roles of polydextrose in food and how it contributes to the texture, taste, and nutritional profile of the products in which it is used. Bulking Agent- Polydextrose is commonly used as a bulking agent in foods that are marketed as "low-calorie" or "reduced-calorie." As a bulking agent, it provides texture and bulk to food products without adding significant calories. This is because polydextrose has a very low calorie content, containing only 1 kilocalorie per gram. For example, polydextrose is often used in reduced-calorie beverages to provide a thicker mouthfeel and improve the overall sensory experience. In baked goods, polydextrose can be used to replace sugar, which reduces the calorie content of the product while maintaining the texture and volume of the finished product. Polydextrose can also be used as a texturizer in food products. It can be used to modify the texture of foods by providing a smooth and creamy mouthfeel, as well as by enhancing the crispness of certain foods. In dairy products such as ice cream and yogurt, polydextrose can be used to improve the texture by providing a smooth and creamy mouthfeel. In baked goods such as cakes and cookies, polydextrose can be used to provide a crispy texture on the outside while maintaining a soft and chewy texture on the inside. Emulsifier and Stabilizer - Polydextrose can also act as an emulsifier and stabilizer in food products. Emulsifiers help to keep oil and water-based ingredients from separating, while stabilizers help to prevent the formation of ice crystals in frozen foods. In salad dressings and mayonnaise, Polydextrose can be used as an emulsifier to keep the oil and water-based ingredients from separating. In ice cream and frozen desserts, polydextrose can be used as a stabilizer to prevent the formation of ice crystals and maintain the smooth texture of the product. Source of Dietary Fiber- Polydextrose is a source of dietary fiber, which is important for maintaining digestive health. Unlike other sources of fiber, such as fruits and vegetables, polydextrose is not digested in the small intestine and is instead fermented by bacteria in the large intestine. This fermentation process produces short-chain fatty acids that provide energy to the cells in the large intestine and promote the growth of beneficial gut bacteria. As a source of dietary fiber, Polydextrose can be used in a variety of food products to increase their fiber content. For example, polydextrose can be added to breakfast cereals, snack bars, and other foods to increase their fiber content and promote digestive health. Low Glycemic Index- Polydextrose has a low glycemic index, which means it does not cause a rapid increase in blood sugar levels. This is because polydextrose is not digested in the small intestine and does not contribute to the rise in blood sugar levels that is typically seen after consuming high-carbohydrate foods. As a low glycemic index ingredient, Polydextrose can be used in foods designed for people with diabetes or those who are trying to manage their blood sugar levels.
0 Comments
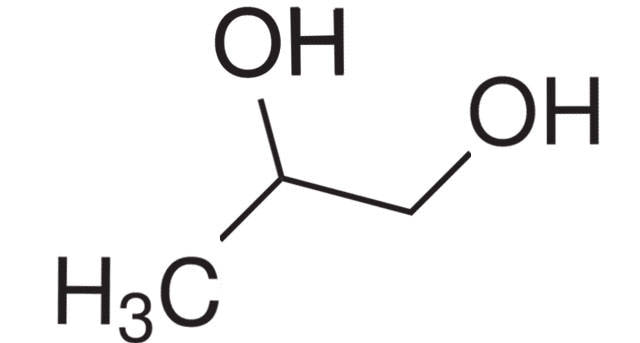
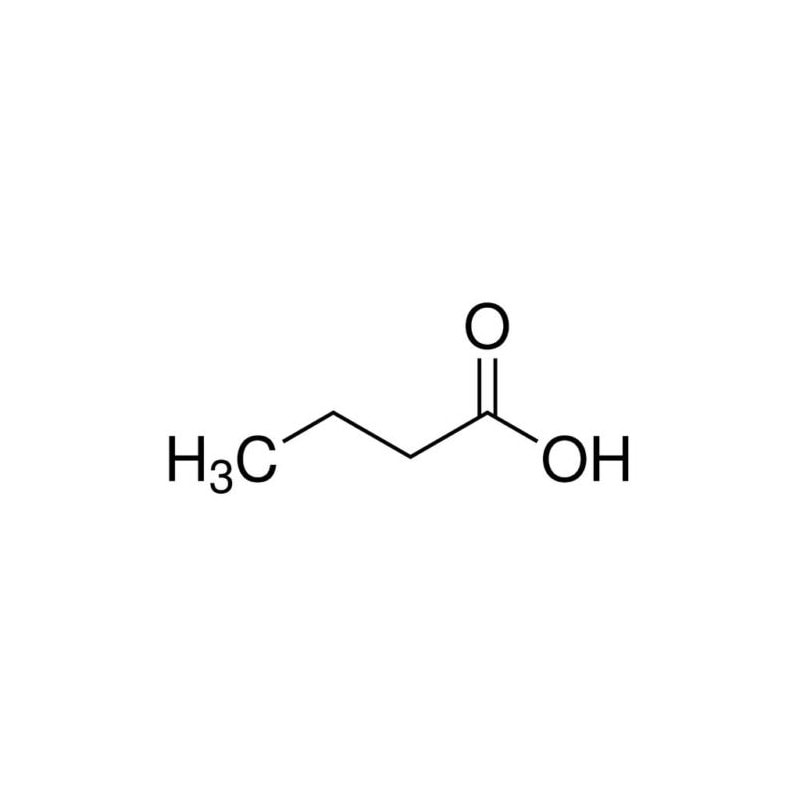
Propylene, also known as propene, is a colorless, flammable gas that is commonly used in a variety of industries for a wide range of applications. It is a member of the alkene family of hydrocarbons and is produced through the cracking of petroleum or natural gas. One of the primary uses of propylene is in the production of polypropylene, which is a type of thermoplastic polymer that is used in a variety of applications, including packaging, textiles, automotive parts, and more. Polypropylene is a versatile and durable material that is resistant to moisture, chemicals, and high temperatures, making it ideal for a range of industrial and commercial uses. Propylene is also used as a feedstock in the production of a variety of other chemicals, including acrylonitrile, propylene oxide, and butyraldehyde. Acrylonitrile is used in the production of synthetic fibers and plastics, while propylene oxide is used in the production of polyurethane foam and other materials. Butyraldehyde is used in the production of various solvents and resins. In addition to its use in the production of various chemicals and materials, propylene is also used as a refrigerant in air conditioning and refrigeration systems. It is also used as a fuel for certain types of vehicles, including propane-powered forklifts and vehicles that run on compressed natural gas. Propylene has a number of properties that make it a useful and versatile chemical. It is highly reactive, which allows it to be easily converted into a wide range of other compounds. It is also relatively stable and can be safely stored and transported under normal conditions. In addition, propylene is non-toxic and has a low environmental impact, making it a popular choice for a variety of applications. While Propylene is generally considered safe when handled and used properly, it can be dangerous if mishandled or exposed to certain conditions. It is a highly flammable gas and can pose a fire or explosion hazard if not properly stored and handled. In addition, it can cause respiratory irritation and other health effects if inhaled in high concentrations. Propylene is a versatile and widely used chemical that is important in a variety of industries and applications. Its primary use is in the production of polypropylene, but it is also used in the production of a range of other chemicals and materials. Propylene is highly reactive and has a number of properties that make it useful for a range of applications, but it can be dangerous if mishandled or exposed to certain conditions. As with any chemical, it is important to handle and use propylene safely and properly to minimize the risks to health and safety. Chlorine derivatives are utilised in a variety of applications, including disinfection and bleaching, organic and inorganic compounds, and metal separation. Chlorine is a highly effective disinfectant that is commonly utilised in water treatment techniques. The expanding household and industrial water treatment industries are likely to drive demand for chlorine in water treatment. Propylene has a three-carbon double bond and is used to make polypropylene resin, acrylonitrile, acrylic acid, propylene oxide, isopropyl alcohol, and acetone via polymerization, oxidation, alkylation, hydration, and halogen addition. Propylene, like ethylene, is an important fundamental chemical in the petrochemical industry. Polypropylene is used in a wide range of applications due to its inexpensive cost and good rigidity, heat resistance, and gloss. It is primarily polymerized using four methods: solution polymerization, suspension polymerization, bulk polymerization, and gas-phase polymerization. Sodium Carbonate has the chemical formula Na2CO3. It is a naturally occurring inorganic substance that is present in large amounts in mineral fluids as well as the solid minerals natron, trona, and thermonatrite. Na2CO3 is a salt that dissolves in water and is white, odourless, and produces moderately alkaline solutions. Sodium carbonate, with the chemical formula Na2CO3, is a diazonium salt of carbonic acid. There is an alkalizing action of sodium carbonate. Sodium hydroxide and carbonic acid are created when sodium carbonate is dissolved in water. Plants growing in sodium-rich soils produce ashes that are collected to make sodium carbonate. Sodium carbonate is also known as soda ash because the ashes of these sodium-rich plants were distinct from the ashes of wood. Using sodium chloride and limestone, the Solvay technique produces a sizeable volume of sodium carbonate (Na2CO3). Large amounts of Sodium Carbonate are created using the Solvay method. The Leblanc process, the electrolytic method, the Hou's process, etc. are only a few examples of other methods that can be used to create it. The most economical of the processes is the Solvay method, also referred to as the ammonia-rich process. The reactants employed in this process include ammonia, brine, and limestone. They are readily available and reasonably priced. In the course of this process, salt could be obtained as a necessary byproduct. This reaction involves the addition of carbon dioxide and ammonia to a cold saturated sodium chloride solution. When sodium ions are present, sodium hydrogen carbonate, which is barely soluble, totally precipitates. In order to produce sodium carbonate, it is filtered out and burned. Large amounts of sodium carbonate are created using the Solvay method. The Leblanc process, the electrolytic method, the Hou's process, etc. are only a few examples of other methods that can be used to create it. The most economical of the processes is the Solvay method, also referred to as the ammonia-rich process. The reactants employed in this process include ammonia, brine, and limestone. They are readily available and reasonably priced. In the course of this process, salt could be obtained as a necessary byproduct. This reaction involves the addition of carbon dioxide and ammonia to a cold saturated sodium chloride solution. When sodium ions are present, sodium hydrogen carbonate, which is barely soluble, totally precipitates. In order to produce sodium carbonate, it is filtered out and burned. A cleaning agent is Sodium Carbonate, sometimes known as washing soda. Salt carbonate is a common ingredient in dry soap powders. It is utilised to get rid of both temporary and enduring water hardness. It is a component in the making of paper, soap, and glass. Borax and other sodium compounds are produced using it. Many culinary uses exist for sodium carbonate. Many applications employ sodium carbonate as a powerful base. In particular, mineral deposits that are created when seasonal lakes evaporate can be found in arid locations naturally and contain sodium carbonate, which is water soluble Alkali Lakes: Sodium Carbonate is present in high concentrations in alkali lakes. The estimated amount of anhydrous sulphate at Soda Lake, California, is close to one million tonnes. Lakes all across the world may contain trace levels of borax. Volcano: Natrite, a mineral type of sodium carbonate that is anhydrous, is very rare. Tanzania's peculiar volcano, Ol Doinyo Lengai, erupts sodium carbonate. Camphor is a natural ingredient found in many topical ointments, balms, and lotions. It helps relieve pain, itching, and eczema by acting as an antispasmodic and antibacterial agent. It can also help reduce the symptoms of some illnesses such as arthritis and rheumatism. However, it should not be taken internally or applied topically without the approval of a health care professional. Camphor has long been used in traditional medicine and herbalism to relieve pain and inflammation, itching, skin infections, spots and blackheads, and respiratory congestion. It is a terpene that comes from the Cinnamomum camphora tree. It has a strong aromatic odor and is valued for its antifungal, antibacterial, and anti-inflammatory properties. Its terpene constituents, including eucalyptol and limonene, have antiseptic and cough-suppressing properties. Inhaled as vapor, it works as an expectorant to clear secretions from the lungs and reduce mucus buildup. It is commonly used in chest rubs to help ease coughing and cold symptoms, and a recent study suggests that it may also work as an effective nighttime cough remedy. It can be applied to the skin to help relieve itching and pain associated with arthritis or rheumatism, and to increase blood flow. However, it should not be rubbed on broken skin or open wounds as it can cause severe irritation and even skin burns. Camphor has anti-inflammatory properties and relieves pain. It is used in a number of medicines to ease joint pains and muscles spasms. It also helps reduce swelling and improves blood circulation to the painful area. It is found in a number of topical creams and ointments, which often contain menthol or eucalyptus oil. A small amount of camphor rubbed into skin helps ease itching and inflammation in atopic dermatitis. Applying a topical cream or ointment containing camphor with menthol and eucalyptus oil can help reduce the size of mosquito bites. Camphor has a strong aroma that helps free the bronchi and other parts of the respiratory system that get blocked with phlegm. This makes it an effective decongestant and is used in a variety of over the counter decongestant balms and cold rubs. It is also a common ingredient in over the counter antihistamines that help treat allergies. It can be applied directly to the skin and is found in many topical products that reduce itching and irritation. Camphor is an essential oil that can be absorbed into the skin and helps improve blood flow, reduce pain and inflammation, and detoxify the body. It can also relieve the itching of dandruff and soothe the scalp. Camphor may also be combined with glucosamine and chondroitin to reduce joint pain in people with osteoarthritis. Camphor is used in a variety of spiritual contexts. It can be burned to purify and cleanse the space, or it can be added to baths or diffusers to help in relaxing and connecting with intuition. It is also a great talisman to ward off evil spirits, malevolent entities, curses, and the evil eye. It can help to clear negative energy and enhance concentration. It is also a natural deterrent for mosquitoes, which are known to spread malaria and dengue fever. This can be beneficial for anyone who is prone to these illnesses, as the scent of camphor drives away mosquitoes that cause them. Propylene is a combination of hydrogen and carbon that is a main feedstock for various business and industrial substances. It is utilized in a diversity of goods, comprising diluters, gums, plasticizer alcohols and makeups. Propylene Glycol is a skin cream, humectant and age reversal cream that aids to inhibit the loss of moisture in the skin. It is a main compound of several cosmetic goods, comprising shampoos and creams, which are equipped to enable the skin look fresher and very glowing. It is a main component of some household cleansers, shampoos and pharma’s. It is dangerous for kids and should be ignored by those who are subtle to it. The most general contact route for a staff is from breath. This is very probable as the gas has an acute half-life, which means that it disperses vastly from the pulmonary while inspired. Anyhow, there is a proof that inhalation may result in damage to the nose outer layer and the breathing epithelium. This can lead to squamous metaplasia of the airlines in the nose and epithelial hyperplasia in the breathing tract. In addition, there is a limited amount of evidence that the chemical can penetrate the skin through direct contact with the surface of the skin or by absorbing from the environment. It is not known whether this is an issue with the substance itself, or if it is a result of improper application of the product to the skin. As a professional risk, staff should be taught in the correct process to detect and react for the processes immediately. Additionally, staff should be skilled in accurate management and disposal of dump and garbage consisting this chemical. Propylene is a usual component in several varied goods. It is a building-block chemical surface for other various composite chemicals and can be seen in a variety of goods comprising packaging film, threads, vessels, flasks and caps. It has several benefits compared to polyethylene that result it to be a unique choice for a vast range of uses. These comprise a huge chemical resistivity, better fatigue potency, and low price production. Additionally, it is Food and Drug Administration approved and 3-A Dairy acquiescent. This causes it to be a great cloth for tank design and constructions that are unprotected to food items, and for keeping chemicals, pharma and lab devices. Other advantage of propylene is that it is not hazardous, making it a protective supplement to utilize in a variety of dishes and other brews. This material is also seen in numerous makeups and skin care goods and aid to stiffen preparations. Propylene has been known to have anti-infectious and anti-bacterial features that can aid decrease acne at start. This aids decrease breaks and inhibit the looks of scarring. It decreases the thickness of some makeup and skin care makings, resulting them spread evenly and enter deeper in the skin. This can be advantageous for individual who have dry skin as it enables the active elements in the goods to enter to the inmost layer of the epidermis, which may enhance their efficacy. The effects of Camphor, a stimulant of the central nervous system (CNS), range from modest CNS excitation to generalised seizures. Camphor is extremely fat soluble and easily passes the blood-brain barrier. Although the exact mechanism of camphor's influence on the oxidation cycle at a phase above the cytochrome b level of the cytochrome oxidase system has not been clarified, it has been proposed that it is intraneuronal. It is primarily a neurotoxic whose chemical composition makes it simple to cross the blood–brain barrier. Besides irritating to skin and mucous membranes, excessive doses of camphor might make you throw up and have diarrhoea. 10% Camphor spirits had been crawled through by a 15-month-old baby. He developed ataxia throughout the course of the following 48 hours and experienced multiple brief generalised motor seizures. Despite receiving appropriate therapy, the seizures persisted for two days. In the course of 15 days, he gradually got better; eventually, his recovery from motor and mental function was whole. Up until a year later, when the mother gave the child a camphorated vaporizer with 4.81% camphor, the youngster had no more seizures. There was a transient significant motor seizure when inhaling. Once distilled from the camphor tree's bark, turpentine oil is now used to make camphor, an aromatic terpene ketone. Several topical and vaporised drugs used to treat musculoskeletal pain or the symptoms of common flu-like illnesses frequently contain it as an active component. Camphor is marketed as an analgesic, an antipruritic, and an antitussive, and it is also included in earlier formulations of mothballs. Whereas its hydroaromatic terpene group's cyclic ketone is thought to be a neurotoxic, the precise method by which Camphor causes toxicity is unknown. Due to its high lipophilicity, camphor moves quickly across cell membranes and is widely distributed. The fact that its metabolites are removed slowly and are retained in fat deposits may be to blame for the delayed onset of seizures brought on by camphor poisoning. 2 Due to its direct impact on mucosal surfaces, camphor may potentially have adverse effects on the digestive system. Seizures and mortality are linked to doses between 750 and 1500 mg, and doses as low as 500 mg in some case reports. 2 Because of this, the U.S. Food and Drug Administration decided in 1982 that goods could only have an 11% maximum camphor content. A teaspoonful of certain commercially available preparations, however, contains 500 mg. A case series of paediatric seizures linked to Camphor toxicity also emphasises the continued use of camphor in several ethnic and cultural rituals. Products with high concentrations of camphor that are illegally sold present a risk to these groups. To improve and preserve the appearance of human skin, Skin Care products are employed. There are many different products on the market, including cleansers, toners, body lotions, oils and serums, and creams with retinol, alpha-hydroxy acids, and beta-hydroxy acids among their main components. Liquid camphor is quickly absorbed by the skin, mucous membranes, and digestive system. Symptoms may start to show up 5 to 90 minutes after intake. Food and other compounds that might affect the rate of camphor absorption are highly correlated with the absorption. The liver converts camphor to a campherol, which is then conjugated with glucuronic acid. It is uncertain if the parent substance, a metabolite, or both are to blame for Camphor toxicity. Metabolites connected to camphor are soluble in fat. As a result, fat tissue may accumulate in substantial amounts. In every tissue, camphor is abundantly dispersed. Within minutes of ingesting between 0.5 and 1.0 g, measurable serum levels become visible. Many crucial uses for butyric acid can be found in the chemical, food, and medicinal industries. The chemical industry uses Butyric Acid extensively to create thermoplastics such cellulose acetate butyrate. Other esters, such as glycerol tributyrate, are crucial in the creation of polymeric polymers. Butyric acid is used to give buttery overtones to food flavours, and its esters are frequently added as food additives to boost fruit aroma. Butyric acid, one of the short-chain fatty acids produced by bacterial fermentation of food fibres in the colon, serves as both a primary source of energy for the human body and a known colon cancer suppressor. Its biological properties, which include therapeutic effects for gastrointestinal disorders, cancer, and hemoglobinopathies, have been extensively researched. The clinical development of a class of acyloxyalkyl butyrate prodrugs is now underway. Furthermore, butyric acid derivatives are utilised to make anaesthetics, antithyroid, and vasoconstrictor medications. Clostridial saccharalyzers convert glucose to Butyric Acid. The starting point will be the intermediate acetyl-CoA. From an energetics perspective, producing acetate as the only byproduct would not be satisfactory since, as the pH falls into the acid range, reoxidizing NADH + H+ becomes more and more challenging. Hence, it is not surprising to discover a cyclic mechanism in the clostridia that is comparable to that in the Propionibacteria. Butyric acid, a byproduct of this cyclic cycle that produces far less acid than acetate, is created. In dry N,N-dimethylformamide, the acid chloride of l-pyrene Butyric Acid, produced in accordance with Fieser and Fieser (1932), was allowed to react with equimolar amounts of pyridine and N,N-dimethyl-ethylendiamine. Six hours were spent conducting the reaction at 65 °C. After adding water, 20% NaOH was used to bring the pH down to 8.5, and diethyl ether was used to partition the liquid. Three times the process was carried out, collecting the organic phase each time. The gathered organic extracts were dried by evaporation. The solid product, which had a melting point of 107–108°C, displayed the proper mass spectrum for 2-[4-(l-pyrenyl-butyrylaminoethyl)]. –diethylamine Valproic acid and Butyric Acid are the two main categories of HDAC inhibitors currently available. They are frequently employed by researchers to better understand how HDAC inhibition works in psychiatric disease models. Because of their broad targeting activity and partly due to their restricted entry into the brain, these two inhibitors typically work at larger doses. The function of certain HDACs in various disorders is unclear due to the lack of research regarding HDAC inhibitory pathways in treating psychiatric disorders. In this context, a greater understanding of the unique functions played by each HDAC may assist to change the practise of HDAC inhibitor therapy. The challenges in producing and purifying the enzymes have hampered attempts to date in developing selective HDAC inhibitors. These enzymes can be purchased commercially, but their high cost prevents widespread use. The fact that not all enzymes process the same substrate presents additional difficulty for drug discovery research teams. A few research teams have attempted to talk about the utilisation of particular substrates for each type of HDAC enzymes. Additional profiling will be required because to the paucity of inhibitory evidence for any inhibitor against all of the HDAC members, which is not frequently documented in the literature. To ensure worker safety and uphold secure working conditions at the workplace, Industrial Gloves are utilized. Natural and synthetic raw materials are both used to make industrial gloves. They can handle low and high temperatures and are resistant to chemical reactions, abrasion, and corrosion. Silicates are a class of minerals that are characterized by their chemical composition and structural properties. These minerals are made up of silicon and oxygen atoms, which are linked together in various configurations to form a wide range of silicate minerals. Silicates are some of the most common minerals found in the Earth's crust, and they play an important role in a variety of geological processes. Chemically, Silicates are compounds that contain silicon (Si) and oxygen (O), with a general formula of SiO4. The SiO4 unit is called a tetrahedron, which consists of a silicon atom at the center and four oxygen atoms arranged in a tetrahedral shape around it. The tetrahedra can be linked together in various ways to form the different silicate minerals. One of the simplest forms of silicates is the mineral olivine, which is composed of isolated SiO4 tetrahedra. In olivine, the tetrahedra are not linked together, but instead, they are stacked on top of one another in a three-dimensional lattice structure. Other silicate minerals, such as pyroxenes and amphiboles, have more complex structures where the tetrahedra are linked together in long chains or sheets. One of the most important characteristics of Silicate minerals is their ability to form rocks. Rocks are aggregates of minerals that are bound together by various forces, such as chemical bonding or physical interlocking. Silicates are the most common mineral group found in rocks, and they play a key role in the formation of many different types of rocks. For example, the igneous rocks that form from the cooling and solidification of magma are mostly composed of silicate minerals. These minerals crystallize from the magma as it cools, and they can form a variety of textures and shapes depending on the conditions of crystallization. Some igneous rocks, such as granite, are composed almost entirely of silicate minerals and are some of the most common types of rock found on the Earth's surface. Silicates also play an important role in the formation of sedimentary rocks. Sedimentary rocks are formed by the accumulation and lithification of sediments, which are small particles of rock or mineral fragments that are transported by wind, water, or ice. Many sediments are composed of silicate minerals, such as quartz, feldspar, and clay minerals, which are resistant to weathering and erosion. These minerals can accumulate over time to form sandstones, shales, and other types of sedimentary rocks. In addition to their role in rock formation, silicate minerals have a wide range of other uses and applications. For example, quartz is a common component of many types of electronics, including computer chips and solar panels. It is also used in the production of glass, as well as in the manufacture of abrasives and gemstones. Other Silicates minerals, such as asbestos, have been used in the past for their heat-resistant and insulating properties. However, the use of asbestos has been largely phased out due to its harmful effects on human health. Silicates minerals also play an important role in the Earth's carbon cycle. Carbon dioxide is one of the most important greenhouse gases that contribute to climate change, and it is removed from the atmosphere through a process called carbon sequestration. One of the primary ways that carbon is sequestered is through the weathering of silicate minerals, which releases calcium and magnesium ions into the soil and water. These ions can react with dissolved carbon dioxide to form bicarbonate ions, which can then be transported and deposited in the ocean, where they become part of the ocean's carbonate sediment. Floor coatings are used to maintain and properly preserve flooring. These add to the aesthetic appeal while providing exceptional resistance to chemicals, abrasion, and impact. Floor Coatings are inexpensive, widely accessible, simple to apply, and offer good surface protection. Aspartame is a popular artificial sweetener that is used as a sugar substitute in a wide range of food and beverage products. The chemical name for aspartame is N-L-α-aspartyl-L-phenylalanine 1-methyl ester. It is commonly used in low-calorie and sugar-free products, including soft drinks, chewing gum, yogurt, and tabletop sweeteners. Aspartame was discovered in 1965 by James M. Schlatter, a chemist working for the pharmaceutical company Searle. He accidentally discovered the sweetening properties of aspartame when he was working on a new anti-ulcer drug. Aspartame was approved for use in the United States by the Food and Drug Administration (FDA) in 1981. The chemical structure of aspartame consists of two amino acids, aspartic acid and phenylalanine, which are linked together by a peptide bond. When ingested, aspartame is broken down into these two amino acids, which are then metabolized by the body. Aspartame is about 200 times sweeter than sugar, and it has a much lower caloric content. This makes it an attractive alternative to sugar for those who are trying to reduce their calorie intake. However, aspartame has been the subject of controversy over the years, with some groups claiming that it is unsafe for human consumption. One of the concerns about aspartame is that it may cause cancer. However, extensive research has been conducted on the safety of aspartame, and there is no evidence to suggest that it is a carcinogen. The FDA, the European Food Safety Authority (EFSA), and other regulatory agencies around the world have all declared aspartame to be safe for human consumption. Another concern about aspartame is that it may cause neurological and behavioral problems. Some people have reported experiencing headaches, dizziness, and other symptoms after consuming products containing aspartame. However, again, extensive research has been conducted on this topic, and there is no evidence to suggest that aspartame causes these types of problems in the general population. In fact, the vast majority of people can consume aspartame without experiencing any adverse effects. Despite the safety of Aspartame being established by numerous studies, some people may still be sensitive to this sweetener. Some people may experience symptoms such as headaches or digestive problems after consuming products containing aspartame. For these individuals, it may be best to avoid aspartame and choose a different sweetener. Aspartame is an artificial sweetener that is used in a wide range of food and beverage products as a sugar substitute. It is much sweeter than sugar and has a much lower caloric content. Food additives are components added to food products to improve the food's texture. Emulsifiers, acidity regulators, enzymes, colourants, flavouring agents, hydrocolloids, preservatives, and sweeteners are some of the several categories that they fall under. Food Additives are frequently used in food products to enhance sweetness, flavour, and nutrients as well as to keep the products fresh and avoid food spoilage. Disinfectants, Any Agent Used To Kill Bacteria On Inanimate Items, Such As Alcohol Or Creosote9/2/2023 Disinfectants are chemical substances used to kill or eliminate harmful microorganisms such as bacteria, viruses, fungi, and parasites from surfaces, objects, and the environment. They play a critical role in promoting public health and preventing the spread of infectious diseases. Disinfectants have been in use for centuries and have evolved over time to meet the changing needs of society. The first known disinfectant was carbolic acid, also known as phenol, which was used in the late 19th century. Today, a wide variety of disinfectants are available, including quaternary ammonium compounds, chlorine-containing compounds, hydrogen peroxide, ozone, and ultraviolet light. Quaternary ammonium compounds, also known as quats, are a common type of disinfectant used in household and commercial settings. They are highly effective against bacteria, viruses, and fungi and are widely used in a range of applications, including sanitizing food processing equipment, cleaning floors and surfaces, and disinfecting medical devices. Chlorine-containing compounds, such as bleach, are another popular type of Disinfectants. These compounds are highly effective against a wide range of microorganisms, including bacteria, viruses, and fungi. They are widely used in hospitals, schools, and other public facilities to disinfect surfaces and objects. Hydrogen peroxide is a powerful oxidizing agent that is effective against bacteria, viruses, and fungi. It is commonly used as a Disinfectants in hospitals, dental clinics, and other healthcare facilities. It is also used as a sanitizer in food processing and agriculture to eliminate harmful microorganisms from fruits, vegetables, and other food products. Ozone is another effective disinfectant that is widely used in water treatment plants, swimming pools, and spa facilities. Ozone is highly reactive and can quickly eliminate microorganisms, including bacteria, viruses, and fungi, from the water. Ultraviolet light is a physical disinfectant that works by destroying the DNA and RNA of microorganisms, effectively killing them. Ultraviolet light is commonly used in air purifiers, water treatment systems, and medical facilities to sterilize surfaces and objects. It is important to note that not all disinfectants are equally effective against all microorganisms, and some may be more effective against certain types of bacteria, viruses, and fungi than others. Additionally, the effectiveness of a disinfectant can be influenced by factors such as the type of microorganism, the concentration of the disinfectant, and the length of exposure time. Disinfectants play a critical role in promoting public health and preventing the spread of infectious diseases. They are available in a wide variety of forms, including quaternary ammonium compounds, chlorine-containing compounds, hydrogen peroxide, ozone, and ultraviolet light. The choice of disinfectant depends on the type of microorganism, the surface or object being disinfected, and the specific application. It is important to use disinfectants properly and according to the manufacturer's instructions to ensure their effectiveness and to prevent any potential harm to humans and the environment. |
Categories
All
|