|
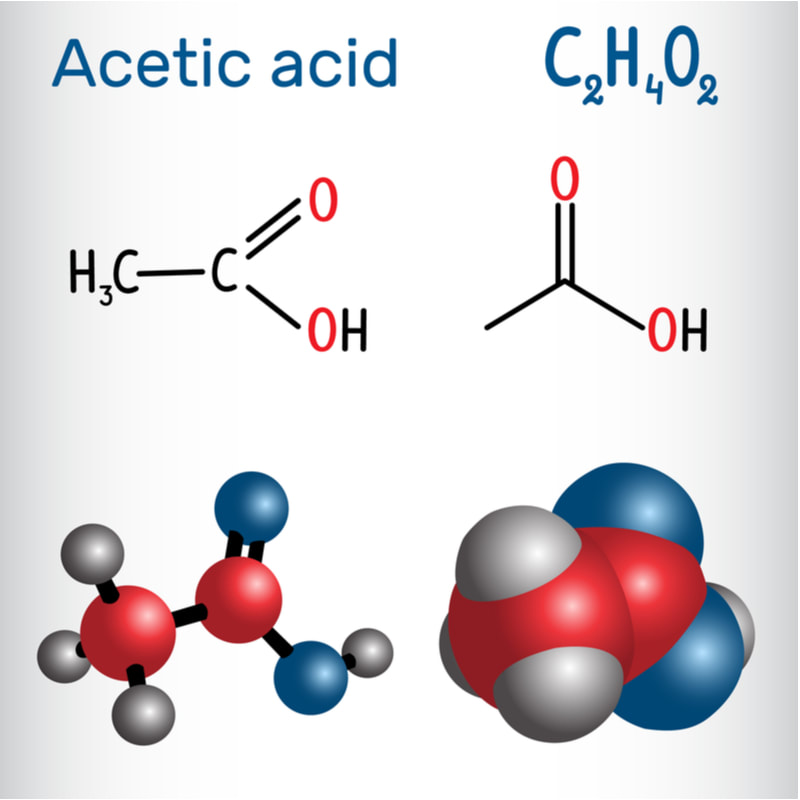
Acetic Acid, a clear, pungent liquid with the molecular formula CH3COOH, plays a vital role in the production of various foods and beverages. Commonly known as the main component of vinegar, acetic acid offers more than just a sour taste. It serves as a versatile ingredient, imparting unique flavors, enhancing preservation, and contributing to the overall quality of numerous food and beverage products. Acetic Acid significantly influences the flavor profile of many food and beverage products. In the culinary world, vinegar, which primarily consists of acetic acid, is used as a condiment, ingredient, and flavor enhancer. Its distinct sour taste adds tanginess and brightness to dressings, sauces, pickles, and marinades. Acetic acid's presence creates a well-balanced flavor profile, heightening taste sensations and complementing other ingredients. A Study By Coherent Market Insights Says, The Global Acetic Acid Market Was Estimated To Be Valued More Than US$ 10,551.4 Million In Terms Of Revenue In 2019 And Is Predicted To Grow At A CAGR Of 9.5% During The Forecast Period (2020 To 2027). Furthermore, acetic acid contributes to the complex flavors in fermented foods and beverages. It acts as a catalyst in fermentation processes, converting sugars into alcohol and subsequently into acetic acid. This transformative process produces a wide range of flavors and aromas, evident in products such as wine, beer, kombucha, and certain types of cheese. Acetic acid's involvement in the fermentation journey brings forth unique sensory experiences, including fruity, floral, or tangy notes, depending on the specific product. Acetic Acid role in food and beverage production is closely intertwined with fermentation. Fermentation is a metabolic process where microorganisms, such as bacteria or yeast, convert sugars into various compounds, including acetic acid. This process occurs naturally or through controlled methods to create specific flavors and preserve foods. In the production of vinegar, the primary source of acetic acid is the fermentation of ethanol. Initially, yeast converts sugars into alcohol during alcoholic fermentation, and subsequently, specific strains of bacteria, such as Acetobacter, oxidize the alcohol to acetic acid in a process known as acetous fermentation. This transformation gives vinegar its characteristic acidity and distinct flavor. Different substrates, such as wine, apple cider, malt, or rice, provide diverse flavors and nuances to the final vinegar product. Apart from vinegar, acetic acid's involvement in fermentation extends to other fermented foods and beverages. For example, in the brewing industry, acetic acid production can be a sign of unwanted bacterial contamination, resulting in off-flavors. However, in controlled scenarios, as in the production of sour beers, specific strains of bacteria intentionally contribute to acetic acid production, adding complexity and tartness to the final product. Acetic Acid possesses antimicrobial properties, making it an effective preservative in food and beverage production. Its ability to inhibit the growth of spoilage-causing bacteria and molds helps extend the shelf life of numerous products. In pickling, acetic acid's presence, either naturally or from vinegar, creates an acidic environment that prevents the growth of harmful microorganisms. This preservation method, known as acidification, helps maintain the quality, texture, and flavor of pickled vegetables and fruits over an extended period. An organic molecule called Vinyl Acetate monomer is created when ethylene and acetic acid are combined with oxygen and a palladium catalyst. It is one of the essential chemical components needed to make a variety of commercial and consumer products, including polymers, which are then used to make coatings, wire, and other building materials, among other things. Furthermore, Acetic Acid preservative properties are valuable in the production of sauces, condiments, and dressings. It inhibits the growth of spoilage microorganisms and contributes to product stability. Acetic acid, along with other natural preservatives, helps maintain the freshness and quality of these products, allowing for longer shelf life and reduced reliance.
0 Comments
Food preservation is a crucial process that aims to extend the shelf life and maintain the quality and safety of food products. Acetic Acid, a naturally occurring organic compound, has been utilized for centuries as a food preservative. Acetic Acid, also known as ethanoic acid, is a weak acid with a pungent odor. It is found naturally in various food sources such as fruits, vinegar, and fermented products. Acetic acid is produced through the fermentation of sugars by acetic acid bacteria. According To Coherent Market Insights, The Global Acetic Acid Market Was Estimated To Be Valued More Than US$ 10,551.4 Million In Terms Of Revenue In 2019 And Is Predicted To Grow At A CAGR Of 9.5% During The Forecast Period (2020 To 2027). Acetic acid acts as an antimicrobial agent, inhibiting the growth of bacteria, yeast, and molds. Its acidic nature disrupts the cellular processes of microorganisms, leading to their inactivation and death. Acetic acid also lowers the pH of food, creating an unfavorable environment for spoilage organisms. Acetic Acid is widely used in pickling processes. It imparts a tangy flavor, extends the shelf life, and inhibits the growth of spoilage microorganisms in pickled vegetables and fruits. Acetic acid is a common ingredient in sauces and condiments like ketchup, mayonnaise, and salad dressings. It enhances the flavor, acts as a preservative, and contributes to the overall stability of these products. Acetic acid is added to canned foods to prevent spoilage and maintain their quality during storage. It inhibits microbial growth and enzymatic activity, preserving the texture, color, and taste of the canned products. Acetic acid is used in baking to control the pH of dough, enhance fermentation, and improve the texture and flavor of baked goods. Acetic Acid can be applied to meat and poultry products to inhibit the growth of bacteria such as E. coli and Salmonella, reducing the risk of foodborne illnesses. An organic molecule called Vinyl Acetate monomer is created when ethylene and acetic acid are combined with oxygen and a palladium catalyst. It is one of the essential chemical components needed to make a variety of commercial and consumer products, including polymers, which are then used to make coatings, wire, and other building materials, among other things. Benefits of Acetic Acid in Food Preservation
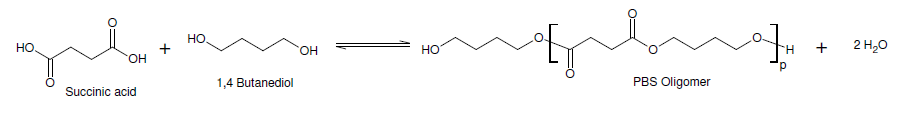
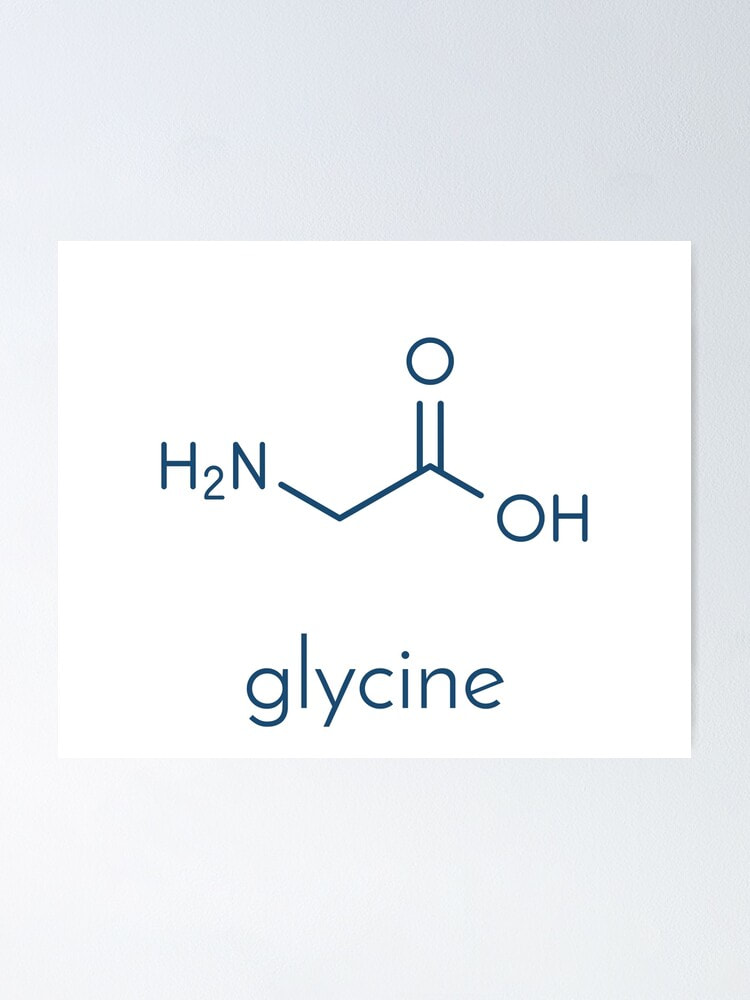
Read the Press Release of Acetic Acid Food is not merely a source of sustenance; it is an experience that tantalizes our senses. From the enticing aroma to the delectable taste and pleasing texture, each element plays a significant role in creating a memorable culinary delight. One ingredient that has gained popularity in the food industry for its ability to enhance taste and texture is Sodium Caseinate. Derived from milk, sodium caseinate offers a range of benefits that contribute to the overall sensory appeal of various food products. Sodium Caseinate is a natural protein derived from milk and is a soluble salt of casein, which is the primary protein in milk. It is obtained by acidifying skim milk, separating the casein, and then neutralizing it with sodium hydroxide. The resulting sodium caseinate is a versatile ingredient that finds application in a wide range of food products. Enhancing Taste: 1. Flavor Binding: Sodium Caseinate possesses excellent flavor-binding properties, allowing it to capture and retain the taste molecules present in food. This characteristic helps in enhancing the overall flavor profile of various dishes, from savory to sweet. It ensures that the taste experience is intensified and more enjoyable. 2. Masking Bitterness: In some food formulations, bitter notes can be a challenge. Sodium caseinate has the ability to mask bitterness, making it an ideal ingredient for improving the palatability of bitter-tasting products, such as certain pharmaceuticals or functional foods. 3. Balancing Saltiness: Sodium caseinate also plays a role in balancing the perception of saltiness in food products. By interacting with salt molecules, it can enhance or reduce the perceived saltiness, providing a more harmonious and well-rounded taste. Enhancing Texture: 1. Emulsification: Sodium caseinate acts as an emulsifier, enabling the blending of two immiscible substances, such as oil and water. This property is particularly valuable in creating smooth and creamy textures in products like salad dressings, ice creams, and sauces. By stabilizing the emulsion, sodium caseinate contributes to a desirable mouthfeel and texture. 2. Foaming and Whipping: Sodium caseinate has foaming properties that help to incorporate air into various food formulations, resulting in a light and fluffy texture. It is commonly used in the production of whipped toppings, mousses, and aerated desserts. 3. Gelling and Binding: The gel-forming ability of sodium caseinate is utilized in the production of gel-based food products, including dairy desserts, custards, and confectionery items. It provides structure and stability, contributing to a pleasant mouthfeel and texture. Applications of Sodium Caseinate: Bakery Products: Sodium caseinate finds application in bakery products like bread, cakes, and pastries. It enhances the texture and extends the shelf life of baked goods, while also contributing to a rich and flavorful eating experience. Dairy Products: In the dairy industry, sodium caseinate is utilized to improve the texture and mouthfeel of products such as yogurt, cheese, and ice cream. It imparts a creamy and smooth consistency, enhancing the overall quality of these items. Beverages: Sodium caseinate is used in the production of various beverages, including coffee whiteners, milkshakes, and protein drinks. It adds creaminess, aids in dispersion, and enhances the sensory attributes of these beverages. Processed Meats: Sodium caseinate is incorporated into processed meat products, such as sausages and deli meats, to improve texture, moisture retention, and binding properties. Alumina Trihydrate is a white, unscented and crystalline synthetic chemical. It is generated from bauxite ore by utilizing the Bayer procedure which includes heating to decrease the aluminium oxides. Alumina trihydrate is also utilized as a flame retardant in the making of plastics and rubbers and also in paints and coats. It is also a prevalent element in antacids as it shields the stomach's pH and is engrossed from the gastrointestinal tract. Manufacturing of Alumina trihydrate leaves a deposit that is usually referred to as red mud. This leftover is a source of natural concern as it has various properties that make disposal very tough. Furthermore, it has several health problems to the individual that come in contact with it because of its high alkalinity and sodicity. The subcommittee presumed that ionized Alumina Trihydrate infuses the epidermis at a rate of 10-3 cm/hr and that it is engrossed at a rate of 0.71 ug/cm2/d. By separating this dermal exposure estimation by the verbal RfD, a non-tumor threat of 0.3 x 10-4 was predicted for alumina trihydrate utilized as a flame retardant in upholstery clothes. A low learning capacity was seen in Long-Evans rats visible orally to 30 or 100 mg aluminum chloride or to 300 mg aluminum trihydrate along with citric acid. Anyhow, the level of consumption for these animals was not assesses and further the background levels for aluminum acceptance could not be detected. There are several benefits of Alumina Trihydrate. That is it has high chemical resistivity, low viscidness, enhanced adhesive standards, and electrical insulating characteristics. It is also a fire retardant and smoke resistivity. It is also non-noxious and halogen free, making it a better substitute to halogenated burn retardants. Alumina Trihydrate is utilized in varied kinds of organizations, such as paper, paints and coatings, and edifice. It is generated from bauxite by the Bayer procedure, which liquefies bauxite in sodium hydroxide at high temperatures. The most considerable progression for Alumina Trihydrate is its usage in the production of paper as an extender pigment. It is very efficient at inhibiting deprivation of the paper goods, and it also includes to its shine. As the attention on sustainability rises, the necessity for alumina trihydrate is known to increase in the upcoming future. Alumina trihydrate is a very helpful filler for polymers, and it can be included to a varied kinds of formulations. It is very helpful in polyester gums as it can enhance the temperature, chemical resistivity, and aging features of the end product. It is also utilized in wire & cable, vinyl, and several thermoset uses also. Alumina Trihydrate is utilized as a stuffing in coatings sector. It has the capability to resist high temperature and provides fire retardant features. It is also halogen-free and low coarse. It is utilized as an antacid and in the therapeutics of canker sores. It can also be utilized as a water permeable substance. Anyhow, the existence of substitutes such as Magnesium Hydroxide can limit the rising usage of alumina trihydrate in the market. Alumino- Alumina Trihydrate is a burn retardant and an insulator. It is a stabilizer for rubber and plastics that are treated at heat below 200 °C. It is also a best filler and extender for elements that can be tarnished by hydrolysis. While open to fire, it endures endothermic dehydration and discharges water vapor that adulterates the ignition gases and toxic fumes. Alumina trihydrate is also utilized in cable sheathing components. It is not engrossed from the skin, however is eaten while swallowed in huge quantities. Orally directed to mice through organogenesis, it did not persuade any notable maternal or evolving properties. Read the Press Release of Alumina Trihydrate Pharmaceutical research constantly seeks innovative solutions to enhance drug development, improve therapeutic efficacy, and mitigate adverse effects. Succinic acid, a naturally occurring dicarboxylic acid, has garnered significant attention as a promising ingredient in pharmaceutical research. Succinic Acid, also known as butanedioic acid, is a colorless, crystalline solid that occurs naturally in various organisms, including plants and animals. It possesses unique chemical and physiological properties that make it an attractive candidate for pharmaceutical applications. Succinic acid is readily available, biodegradable, and exhibits low toxicity, making it environmentally friendly and safe for use in medicinal products. In living organisms, succinic acid plays a vital role in energy metabolism. It is an intermediate in the tricarboxylic acid (TCA) cycle, also known as the Krebs cycle or citric acid cycle, where it participates in ATP production through oxidative phosphorylation. This metabolic involvement suggests that succinic acid may have an impact on cellular processes and signaling pathways, making it an intriguing target for pharmaceutical research. Inflammation is a hallmark of numerous diseases, including autoimmune disorders, cardiovascular conditions, and neurodegenerative disorders. Succinic Acid has demonstrated anti-inflammatory properties by modulating the expression of pro-inflammatory cytokines and inhibiting the activation of NF-κB, a key regulator of inflammation. These findings suggest that succinic acid could be utilized in the development of novel anti-inflammatory drugs. Oxidative stress, resulting from an imbalance between free radicals and antioxidants, contributes to the pathogenesis of several diseases, including cancer, diabetes, and neurodegenerative disorders. Succinic acid possesses antioxidant properties, neutralizing free radicals and protecting cells from oxidative damage. By scavenging reactive oxygen species, succinic acid may have potential applications in the prevention and treatment of oxidative stress-related conditions. Neurodegenerative disorders, such as Alzheimer's and Parkinson's diseases, pose significant challenges to global healthcare systems. Succinic Acid has demonstrated neuroprotective effects by attenuating neuronal damage, reducing oxidative stress, and modulating neurotransmitter levels. These findings highlight the potential of succinic acid as a therapeutic agent for neurodegenerative diseases. In pharmaceutical research, succinic acid has shown promise as an ingredient in drug delivery systems. Its hydrophilic nature and ability to form complexes with various drugs enable controlled drug release, improving bioavailability and therapeutic efficacy. Additionally, Succinic Acid can enhance the stability of formulations and aid in the solubilisation of poorly soluble drugs, addressing formulation challenges in drug development. Cancer remains a global health concern, necessitating the development of effective therapies. Succinic acid exhibits anti-cancer properties by inhibiting tumor cell growth, inducing apoptosis, and suppressing angiogenesis. Moreover, succinic acid-based prodrugs have been investigated to enhance drug delivery and target cancer cells specifically. These findings suggest that succinic acid holds promise as a valuable component in anti-cancer strategies. The polycondensation process between polyalcohols and polyacids results in polyester resin. Unsaturated polyester resins are typically utilised in conjunction with strengthening components like glass fibre. Unsaturated Polyester Resin is extensively employed in a variety of sectors, including marine, construction, electrical, and transportation. Succinic Acid has demonstrated potential in wound healing and tissue regeneration applications. It promotes fibroblast proliferation, collagen synthesis, and angiogenesis, thereby accelerating wound closure and tissue repair. By harnessing the regenerative properties of succinic acid, researchers aim to develop innovative approaches for wound management and tissue engineering. Silicates are used in many applications from abrasives to glass and refractory bricks. They can be found naturally and synthetically. They are also widely used as a binder and cement. Single-chain silicates link together by sharing oxygen ions. This is a common structure in minerals like olivine and garnet. Double-chain silicates link together with a wider range of cations. Silicon is the eighth most common element in the universe and it is found in nature in a variety of forms. It is a hard material, whereas when mixed with water it becomes a soft substance that can be molded into various shapes. It is also used in a variety of different manufacturing, technological and artistic applications. The main Silicates structural unit is a tetrahedral silicon atom linked to four oxygen atoms. These tetrahedra can be linked together to form polymers called siloxane. They can also be joined to one, two or three tetrahedra through shared oxygen atoms. This results in a sheet-like structure with weak bonding forces between the silicate sheets. Sodium silicate is used as a deflocculant in clay slips, usually in combination with soda ash. It is also commonly used in the creation of crackled texture in wheel-thrown pottery. Sodium silicate attacks the plaster in molds much more than other more modern deflocculants and it is difficult to over-deflocculate a slip with it. Refractory bricks are used in high temperature environments, such as in furnaces. They are made from alumina and silica, the oxides of aluminum and silicon. These materials can withstand extreme temperatures and are ideal for use in ceramic-, glass-, and brick-making. They can also be combined with other elements to create new materials. Using these materials will increase the efficiency of the heating process and improve energy conservation. These materials have a low melting point and can be mixed easily with water. Silicates are used for construction projects, especially in high-rise buildings and homes. They provide insulation and prevent heat loss. These bricks are also good for lining furnaces and other heating equipment. Silica refractories are often classified by their mineral composition and degree of transformation. The crystalline modification of silica can be transformed reversibly from its high to its low temperature form, which can affect the density of the material. During firing and slow cooling, these changes in the volume can cause considerable stress within the structure of the bricks. Abrasives are used in a variety of industrial applications including sand blasting, cutting stone, machining, grinding, polishing, and cleaning. The abrasive material can be natural or synthetic, and it is usually coated to make it suitable for use with an abrasive tool. Most of the abrasives available are natural, such as corundum and emery, but some are synthetic, such as silicon carbide and alumina. Other abrasives are called super abrasives and include diamond and cubic boron nitride. Silica is a very common abrasive material and can be found in a variety of minerals and rocks. Examples of Silicates mineral include sand, fused silica, quartz, feldspar, flint, koalin, pumice, and clay-based materials. These mineral silicates are known as rock-like silicates or metasilicates. In these materials, the silicon atom sits in the center of an idealized tetrahedron with oxygen atoms at the corners. The bonds between silicate anions are strong and crosslink to form a rigid structure. Silicates are used to make a variety of laboratory apparatus including prisms, eyeglasses, and cuvettes. These glasses are able to withstand high temperatures and are useful for many analytical applications. However, the dissolution of silicates from glassware can contaminate water samples and reagents. The kinetics of silicate dissolution are dependent on contact time, salinity and pH, as well as the size and shape of the container. Soda Ash Is An Important Chemical Compound That Is Used In A Variety Of Industrial Processes3/5/2023 Soda Ash, also known as sodium carbonate, is an essential chemical used in a wide range of industries, including glass, chemicals, detergents, and pulp and paper. The global demand for soda ash is expected to continue to increase, driven by factors such as population growth, urbanization, and economic development. However, the production of soda ash has significant environmental impacts, including greenhouse gas emissions, water consumption, and land use. The production of Soda Ash is an energy-intensive process that generates significant amounts of greenhouse gas emissions. The two primary methods of soda ash production are the Solvay process and the Trona process. The Solvay process involves the reaction of limestone (CaCO3), salt (NaCl), and ammonia (NH3) to produce soda ash (Na2CO3). The Trona process involves the mining and processing of trona ore, a naturally occurring mineral containing sodium carbonate. Both processes rely on the combustion of fossil fuels, primarily natural gas, to generate the high temperatures needed for chemical reactions. The combustion of natural gas releases carbon dioxide (CO2) into the atmosphere, contributing to climate change. According to the Intergovernmental Panel on Climate Change (IPCC), the industrial sector, which includes soda ash production, accounted for approximately 37% of global CO2 emissions in 2018. The soda ash industry is estimated to be responsible for approximately 1% of global CO2 emissions, with the majority of these emissions coming from the combustion of natural gas. The production of Soda Ash also requires significant amounts of water. The Solvay process relies on the use of brine, a concentrated solution of saltwater, as a feedstock. The Trona process requires large volumes of water for mining and processing trona ore. Both processes also require water for cooling and steam generation. The water used in soda ash production is often saline, which can be challenging to treat and reuse. Water scarcity is a growing concern in many parts of the world, particularly in arid and semi-arid regions where soda ash production is often located. The World Resources Institute estimates that 17 countries, home to one-quarter of the world's population, face "extremely high" water stress. In these regions, water scarcity can lead to conflicts between different water users and exacerbate social and economic inequality. The production of Soda Ash also has significant impacts on land use. The Trona process involves the mining of trona ore, which can have adverse effects on soil quality and vegetation. The Solvay process requires the mining of limestone, which can lead to the destruction of natural habitats and ecosystems. Furthermore, the storage and disposal of waste generated during soda ash production can also have significant impacts on land use. The Solvay process generates calcium chloride (CaCl2) as a byproduct, which can be challenging to dispose of safely. The Trona process generates waste products such as tailings and brine, which can also pose environmental risks if not managed properly. Soda Ash production has significant environmental impacts, including greenhouse gas emissions, water consumption, and land use. As the demand for soda ash continues to grow, it is essential to explore strategies to mitigate these impacts. Some of these strategies include improving the efficiency of soda ash production processes, increasing the use of renewable energy sources, implementing water conservation measures, and improving waste management practices. Garbage Disposals are beneficial to the environment, and one additional benefit is that the end product created after waste is processed is utilised to create biofuels. It is employed in both industrial and domestic settings. On the basis of the type of product being disposed of, garbage disposals are further divided into batch and continuous feed disposals. Governments, industries, and consumers can all play a role in reducing the environmental impact of soda ash production. Governments can incentivize the adoption of cleaner production methods and regulate emissions and waste disposal practices. Industries can invest in research and development to improve the efficiency and sustainability of soda ash production. Sodium Percarbonate is a chemical compound that is widely used as a cleaning and bleaching agent. It is a white, crystalline powder that is composed of sodium carbonate and hydrogen peroxide. When dissolved in water, it releases hydrogen peroxide, which acts as a powerful oxidizing agent. Sodium percarbonate is a versatile compound that can be used in a wide range of applications, from laundry detergents to disinfectants. One of the primary uses of Sodium Percarbonate is as a laundry booster and bleach. It can be added to laundry detergents to help remove stains and brighten fabrics. When dissolved in water, sodium percarbonate releases oxygen, which helps to break down stains and dirt. It is particularly effective at removing stubborn stains, such as coffee, wine, and blood. Sodium percarbonate is also a safer alternative to chlorine bleach, as it does not release harmful fumes and is less likely to damage fabrics. In addition to its use in laundry detergents, sodium percarbonate is also used in household cleaning products. It can be added to all-purpose cleaners, bathroom cleaners, and disinfectants to help remove stains and kill bacteria. Sodium percarbonate is effective at removing mold and mildew, as well as disinfecting surfaces. It is also a safer alternative to chlorine bleach, as it does not release harmful fumes. Sodium Percarbonate is also used in industrial applications, such as in the production of paper and textiles. It can be used as a bleaching agent to whiten paper pulp and textile fibers. Sodium percarbonate is also used in the production of detergents and cleaning products for commercial and industrial use. One of the advantages of sodium percarbonate is that it is environmentally friendly. Unlike chlorine bleach, which can release harmful fumes and contribute to air and water pollution, sodium percarbonate breaks down into oxygen, water, and soda ash. It does not harm aquatic life or contribute to the depletion of the ozone layer. Sodium percarbonate is also a safer alternative to chlorine bleach for use in the home. Chlorine bleach can release harmful fumes and irritate the eyes and lungs. Sodium percarbonate, on the other hand, is less likely to cause irritation and does not release harmful fumes. Sodium Percarbonate is also easy to use. It is available in a white, crystalline powder form that can be easily dissolved in water. It can be added to laundry detergents or household cleaners to boost their cleaning power, or used on its own as a bleach or disinfectant. It is also affordable and readily available, making it a popular choice for both household and commercial use. Another advantage of sodium percarbonate is its effectiveness in removing mold and mildew. It can be used to disinfect surfaces and kill bacteria, making it a popular choice for use in bathrooms and kitchens. It is also effective at removing odors, making it a popular choice for use in pet areas or in the home of smokers. However, Sodium Percarbonate should be handled with care, as it can be harmful if ingested or inhaled. It should be stored in a cool, dry place away from heat and moisture. It should also be kept out of reach of children and pets. Sodium percarbonate is a versatile chemical compound that is widely used as a cleaning and bleaching agent. It is effective at removing stains and disinfecting surfaces and is a safer and more environmentally friendly alternative to chlorine bleach. Sodium percarbonate can be used in a wide range of applications, from laundry detergents to industrial cleaning products. However, it should be handled with care and stored properly to avoid accidents or harm. Glycine is a category of amino acid. The body can produce glycine in its own, however, it is also taken in the form of diet. The sources of food from which one can obtain glycine are fish, diary, meat and legumes. Glycine is a building block for creating the proteins in the body. Glycine is also included in the changing and converting of the chemical signals and the indications in the brain, hence there’s interest in utilizing it for the schizophrenia and the enhancing memory. A typical diet comprises of around 2 grams of the glycine regularly. People utilize glycine for the schizophrenia, memory, stroke, insomnia and the thinking skills and various other purposes and situations, however, there is no better and correct scientific proofs and reasons to assist the utilizations. Schizophrenia –taking the glycine orally additionally with some of the medicines that are antipsychotic looks to decrease some specific symptoms that are the negative prodromes, in few individuals who do not respond to the therapy with the conventional medicines however, it is not yet evident and proved that Glycine is assisted to aid people who are the new medicines such as clozapine. There is an interest in utilizing the glycine for various purposes however, there is no particular safe information and data which could state whether it is helpful or not. When it is taken orally there are chances to be secured for many individuals when is taken in the regular dosages of 6 grams for a month without fail. It is generally well-tolerated, few people might face some of the complications such as the stomach upset. When glycine is applied to the skin there is no certain information to identify whether is better or has any complications. People who are pregnant or are breast feeding then they are advised not to take due to no particular information. The US PNF standards declare that the quantity of any individual impurity in glycine should not surpass 0.1% Collagen, a protein that gives assistance for the bones and other cells and organs in the entire body. Survey and studies state that Glycine can enhance sleep quality in individuals with insomnia and other sleep diseases. It has been known to rise serotonin levels and to aid one get into a deep, very restful sleep cycle. It looks that the hippocampus, a portion of the brain accountable for learning and memory, is triggered while one take glycine additives. This can aid enhance the daytime cognitive working, particularly in young people. Glycine can aid decrease infection by inhibiting oxidative stress. It lessens infectious cytokines such as TNF-alpha, IL-6, and IL-8 while one consume it orally. In animal designs, it also aids decrease liver damage happening due to consumption of alcohol, which can cause alcoholic cirrhosis and fatty liver. Survey show that a high volume of Glycine in the plasma is associated to a decreased threat for cardiovascular disorders and stroke, and to a reduced threat of type 2 mellitus. It can also aid neutralize blood sugar, which can cause fewer sugar cravings and several others. Glycine is also a herald of glutathione, the main flavonoid that combat over free radicals. As people know, glutathione levels originally fall and glycine can aid to reinstate them. Glycine is an amino-acid that is rich in protein and that can further be utilized as a neurotransmitter. This states that it has a direct effect on the brain, and is accountable for numerous problems related to brain. Studies have reported that glycine decrease the threat of coronary heart disorder and acute myocardial infarction. In addition, it may aid to decrease the threat of developing type 2 mellitus and fatness/ obesity. Propylene, also known as propene, is a colorless and flammable gas that is commonly used in a variety of industrial applications. It is a hydrocarbon with the chemical formula C3H6 and is a byproduct of the refining of crude oil and natural gas. In regions such as the United States, the high expansion of industries has increased the requirements for propylene. Propylene is an important building block in the petrochemical industry and is used to produce a wide range of products including plastics, rubber, solvents, and other chemicals. It is also used as a fuel gas for heating and cooking, and as a refrigerant in air conditioning systems. One of the most important uses of Propylene is in the production of plastics. Propylene is used to produce a variety of plastics, including polypropylene, which is commonly used in packaging, textiles, and automotive parts. Polypropylene is lightweight, durable, and has a high resistance to heat, making it a versatile material for a wide range of applications. Propylene is also used in the production of synthetic rubber. It is a key ingredient in the production of ethylene-propylene-diene monomer (EPDM) rubber, which is used in a variety of applications, including automotive parts, roofing, and electrical insulation. Propylene is used as a solvent in a variety of applications, including in the production of paints, coatings, and adhesives. It is also used as a solvent for cleaning and degreasing industrial equipment. Propylene is used as a starting material for the production of a wide range of chemicals, including propylene oxide, acrylonitrile, and cumene. These chemicals are used in the production of a variety of products, including coatings, plastics, and synthetic fibers. Propylene is a flammable gas and can be hazardous if not handled properly. It is important to store and handle propylene following safety regulations and guidelines. It is also important to use appropriate personal protective equipment when working with propylene, including gloves, safety glasses, and a respirator. Exposure to high levels of propylene can irritate the eyes, nose, and throat. Prolonged exposure to propylene can also cause dizziness, headache, and nausea. Ingestion of propylene can cause abdominal pain, nausea, and vomiting. |
Categories
All
|