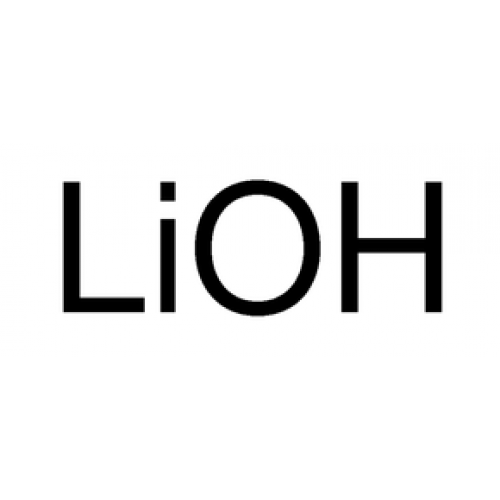|
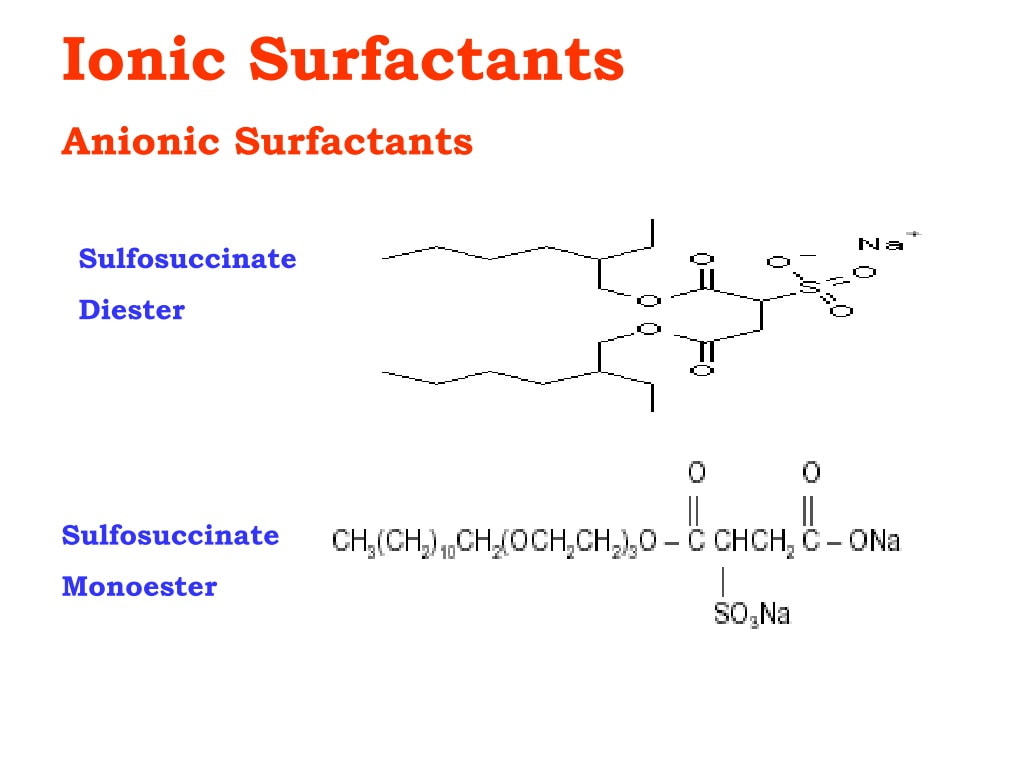
Anionic Surfactants play a pivotal role in various cleaning applications, ranging from household cleaning products to industrial processes. They are known for their excellent cleaning properties, surface activity, and versatility. In recent years, significant advancements have been made in the field of anionic surfactants, leading to enhanced cleaning performance and improved environmental sustainability. This article aims to explore these advances, highlighting their impact on cleaning efficiency, formulation flexibility, and environmental considerations. One of the key areas of advancement in anionic surfactants is their enhanced cleaning performance. Researchers have focused on developing surfactants with superior solubilization and wetting abilities, enabling them to penetrate and remove dirt, grease, and stains more effectively. For instance, the introduction of branched alkyl chains in Anionic Surfactants has resulted in improved oil removal capabilities. Furthermore, modifications to the surfactant structure, such as incorporating hydrophobic and hydrophilic groups, have led to increased surfactant efficacy across a wide range of cleaning tasks. Another aspect of improved cleaning performance lies in the development of tailored anionic surfactant blends. By combining different types of anionic surfactants, such as alkyl sulfates, alkyl ether sulfates, and linear alkylbenzene sulfonates, formulators can achieve synergistic effects, enhancing detergency and soil removal. Additionally, the use of co-surfactants and additives has further contributed to improved cleaning efficiency. Co-surfactants, such as nonionic or cationic surfactants, can enhance the stability and performance of anionic surfactant-based formulations, especially in challenging cleaning conditions. Advances in Anionic Surfactants have also expanded the formulation flexibility for manufacturers. Traditional anionic surfactants were primarily limited to alkaline pH ranges due to their sensitivity to acidic conditions. However, recent developments have led to the creation of acid-stable anionic surfactants, allowing for the formulation of acidic cleaning products. This flexibility enables the development of cleaners that are effective on specific types of soils or surfaces that require lower pH levels for optimal performance. Furthermore, the compatibility of anionic surfactants with various additives and builders has improved. This compatibility enables formulators to incorporate additional ingredients, such as enzymes, polymers, and chelating agents, into cleaning formulations without compromising the surfactant's performance. These additives can enhance specific cleaning properties, such as stain removal, scale inhibition, and foam control, further expanding the range of applications for anionic surfactant-based cleaners. In recent years, there has been an increased focus on developing sustainable cleaning solutions, and anionic surfactants have not been exempt from this trend. Advances in anionic surfactants have led to the development of more environmentally friendly options. For instance, the introduction of bio-based anionic surfactants derived from renewable resources has reduced the reliance on fossil fuel-based feedstocks. Additionally, improvements in the biodegradability and aquatic toxicity of anionic surfactants have resulted in formulations that are more environmentally benign. Every home needs flooring and carpeting, and they can also be employed in other industries including aerospace, vehicles, and aircraft. Wood, vinyl sheets, ceramic tiles, carpets, rugs, and polymer staple fibers are the main components of Flooring And Carpets. Significant advances in Anionic Surfactants have led to enhanced cleaning performance, increased formulation flexibility, and improved environmental sustainability. The development of surfactants with superior cleaning capabilities, the use of tailored surfactant blends, and the improved compatibility with additives have all contributed to the progress in cleaning efficiency. Furthermore, the focus on environmental considerations has resulted in the development of bio-based and more biodegradable anionic surfactants. As the demand for effective and sustainable cleaning solutions continues to grow, the advancements in anionic surfactants are poised to play a significant role in meeting these needs.
0 Comments
Soda Ash, also known as sodium carbonate (Na2CO3), is a versatile chemical compound that plays a vital role in various industries, particularly in the production of glass. With its unique properties and applications, soda ash serves as a critical intermediate in the manufacturing processes of numerous products. Glass is one of the oldest and most widely used materials in human history. It is a solid material, typically transparent or translucent, that is made by melting a mixture of silica (SiO2), soda ash (Na2CO3), and lime (CaO) at high temperatures. Soda ash acts as a fluxing agent in glass manufacturing, facilitating the melting of silica and reducing the melting point of the mixture. This characteristic is crucial in the production of glass because it allows the glass-making process to occur at lower temperatures, making it more energy-efficient and cost-effective. The production of glass begins with the raw materials, primarily silica sand, Soda Ash, and limestone. These materials are carefully selected and processed to ensure the desired glass composition. Soda ash, as an essential ingredient, contributes to the chemical and physical properties of the final glass product. Firstly, soda ash acts as a flux, lowering the melting point of the glass mixture. By reducing the melting temperature, it enables the glass to be manufactured using less energy and shorter processing times. This energy efficiency not only reduces production costs but also has environmental benefits by minimizing the carbon footprint associated with glass manufacturing. Moreover, soda ash plays a crucial role in controlling the viscosity of molten glass. Viscosity refers to the resistance of a liquid to flow. In the glass-making process, controlling the viscosity is essential for shaping and forming the glass into desired products, such as bottles, windows, or glassware. Soda ash helps regulate the viscosity by balancing the ratio of network-forming oxides (such as silica) to network-modifying oxides (such as soda ash) in the glass composition. This balance ensures that the molten glass maintains the desired consistency for shaping and molding. Furthermore, Soda Ash enhances the durability and chemical resistance of glass. Glass produced with soda ash exhibits improved resistance to chemical corrosion, making it suitable for various applications. This property is especially important in industries where glass comes into contact with corrosive substances or environments, such as chemical processing plants, laboratories, and the automotive industry. Soda ash also contributes to the transparency and clarity of glass. It helps reduce the formation of bubbles and other impurities during the melting process, resulting in a clear and transparent final product. This quality is crucial in applications where optical clarity is required, such as in the production of high-quality flat glass for windows or optical lenses. In addition to its role in glass production, soda ash finds extensive use in other industries and applications. For instance, it is a vital component in the manufacturing of detergents and soaps. Sodium carbonate acts as a water softener, helping to remove mineral deposits and enhance the cleaning efficiency of detergents. It is also used as a pH regulator in various chemical processes, including water treatment, textile manufacturing, and pulp and paper production. Soda Ash is utilized in the metallurgical industry for metal refining and as a flux in the extraction of certain metals from their ores. It is also a key ingredient in the production of chemicals, such as sodium bicarbonate (baking soda), sodium silicates, and sodium phosphates, which have numerous applications in food processing, pharmaceuticals, and water treatment. Get the Press Release of Soda Ash UV coatings have emerged as a crucial technology in the field of surface protection, offering enhanced durability and performance to a wide range of products. These coatings utilize the power of ultraviolet (UV) light to create a robust and long-lasting protective layer on various surfaces, including wood, plastics, metals, and composites. At the core Of UV Coatings is the principle of photochemical reaction. UV coatings are composed of specially formulated resins, monomers, photoinitiators, and additives. When exposed to UV light, these coatings undergo a rapid curing process, transforming from a liquid or semi-liquid state into a solid, cross-linked film. This curing process is facilitated by the photoinitiators, which absorb the energy from the UV light and initiate a chain reaction that leads to the formation of strong chemical bonds. The ability of UV coatings to cure rapidly is one of their key advantages. Unlike traditional coatings that rely on heat or chemical reactions for curing, UV coatings cure almost instantaneously upon exposure to UV light. This enables faster production cycles, improved efficiency, and reduced energy consumption. Additionally, the rapid curing process eliminates the need for solvent evaporation, reducing volatile organic compound (VOC) emissions and making UV coatings an environmentally friendly choice. The cross-linking of molecules during the curing process gives UV Coatings their exceptional durability. The resulting film is highly resistant to abrasion, chemicals, and physical impact. This durability ensures that the coated surfaces can withstand harsh conditions, extending the lifespan of products and reducing the need for frequent maintenance or replacement. Another key aspect of UV coatings is their ability to provide superior adhesion. The strong chemical bonds formed during the curing process create a tight bond between the coating and the substrate, ensuring long-term adhesion even under challenging circumstances. This adhesion strength is particularly crucial for applications where the coated surfaces are exposed to moisture, temperature fluctuations, or mechanical stresses. UV coatings also offer excellent clarity and transparency, making them ideal for applications where aesthetics play a significant role. The cured coatings exhibit minimal yellowing or discoloration over time, allowing the underlying surfaces to maintain their original appearance. This is particularly important for products such as automotive components, electronic devices, and furniture, where visual appeal is paramount. In addition to enhancing durability, UV Coatings provide additional functional benefits. They can offer protection against UV radiation, which can be detrimental to the integrity and appearance of surfaces. UV coatings can act as a barrier, blocking harmful UV rays and preventing degradation, fading, or yellowing of underlying materials. This UV protection is highly advantageous in outdoor applications or for products exposed to natural or artificial light sources. Furthermore, UV coatings can incorporate various additives to impart specific properties to the coated surfaces. These additives can provide scratch resistance, anti-fogging properties, anti-static properties, or even antimicrobial properties, depending on the desired application. This versatility allows UV coatings to meet the specific requirements of different industries and enhance the functionality of products beyond basic surface protection. Paper bags, corrugated boxes, clamshells, paper bags, paper cartons, carton & folding boxes, cups & trays, tapes & labels are examples of the flexible and rigid paper packaging that is available in a variety of different configurations. Paper Packaging offers significant weight-related benefits that provide long-term savings on raw materials and transport costs. It is worth noting that the successful application of UV Coatings requires careful consideration of several factors. These include the selection of appropriate coating formulations, proper curing equipment, and optimal process parameters. Factors such as UV light intensity, exposure time, and coating thickness must be precisely controlled to achieve consistent and desirable results. Read Press Release of UV Coatings Silicates is a crystalline mineral composed of silicon dioxide. It is the main component of sand and plays a critical role in many products. Glass, ceramics, abrasives, concrete, metal production and construction are just a few of the ways in which silica is used. Calcium carbonate is one of the most common compounds on the earth. It occurs in aragonite, calcite, limestone, travertine, and other rocks. It is also found in animal bones and shells. It is a major building material, and it is used in many industries including metallurgy, glass, alkali making, rubber, medicine, pigments, and organic chemicals. Calcium carbonate is a natural chemical compound and is widely used as a filler in various plastics. It is also a popular extender pigment in paints and coatings. It can be used to increase the gloss, opacity, and hiding power of the paint and has reinforcing properties. Silicates bricks are a type of refractory material used in manufacturing. They can withstand high temperatures and can be used in a variety of applications. These refractory bricks can be manufactured in different sizes and shapes. They are also available in many different colors and designs. They are a type of refractory that can be manufactured with silica ore, mineralizers, and binders. They can be shaped into the desired shape and fired in a kiln. These refractory bricks are used in a number of industries, including metal fabrication and the manufacture of ceramics. They are available in a range of different sizes and can be purchased at affordable prices. Silicates are a group of minerals that contain silicon and oxygen atoms. They are commonly used in manufacturing as abrasives. There are three different types of silicates: Inosilicates, Phyllosilicates and Tectosilicates. Inosilicates are single-chain silicates that share two bridging oxygen atoms with each silicon atom. Phyllosilicates are a type of silicate that have a sheet structure. In a phyllosilicate, three tetrahedra oxygen are shared with each silicon atom. These silicates have a negative charge per tetrahedral oxygen. This negative charge is neutralized by cations that exist between the tetrahedra of the silicate. Examples of phyllosilicates include clay, mica and talc. Laboratory equipment, including instruments, utensils, and chemicals, is necessary for conducting scientific experiments. It also helps keep labs organized and efficient. Lab equipment can include general tools, such as computers and freezers, or specialized equipment, such as cell culture workstations or microscopes. Regardless of the type, laboratory equipment is crucial to conducting precise tests and research. Volumetric flasks are commonly used in chemist’s laboratories and school laboratories as they help measure liquids accurately. They are made from glass and are calibrated to hold a precise volume of liquids at any given temperature. The uses of Silicates are diverse, and range from a wide variety of construction products to food preservation, metal casting, and paint and adhesives. Silica is a naturally occurring compound in the earth that can be found in crystalline forms, such as anhydrous, tridymite, cristobalite, and quartz. There are numerous silicas, and the primary types include sodium silicate, calcium silicate, zirconium silicate, potassium silicate, magnesium silicate, and ethyl silicate. Each type of silica has a specific set of properties that are required for different applications. Sodium silicate is used as a base or feedstock for formulated and derivative products such as detergents, metals, glass cleaning, and silica gel. Defibrillators are medical devices used to treat life-threatening cardiac arrhythmias, which are abnormal heart rhythms that can lead to cardiac arrest. Defibrillators work by delivering an electrical shock to the heart, which can restore normal heart rhythm and save a person's life. There are two main types of defibrillators: external and internal. External defibrillators are typically found in hospitals, emergency rooms, and on ambulances, while internal defibrillators are implanted directly into the body and are used to treat chronic heart conditions. External Defibrillators work by delivering a controlled electrical shock to the heart through a set of pads placed on the patient's chest. These pads contain electrodes that detect the patient's heart rhythm and deliver an appropriate shock if necessary. The defibrillator is controlled by a medical professional, who monitors the patient's heart rhythm and determines when a shock is needed. Internal defibrillators, on the other hand, are implanted directly into the body and work automatically to detect and treat abnormal heart rhythms. These devices are typically used to treat people with chronic heart conditions, such as arrhythmias that are resistant to other forms of treatment. The basic principle of defibrillation is to deliver an electrical shock to the heart, which depolarizes a critical mass of the heart muscle, stops the arrhythmia, and allows normal sinus rhythm to be reestablished by the body's natural pacemaker in the sinoatrial node of the heart. The energy delivered to the heart is measured in joules, which is a unit of energy. The amount of energy required to successfully defibrillate a patient depends on several factors, including the patient's size, the location of the electrodes, and the type of arrhythmia being treated. During a defibrillation procedure, the patient is typically sedated and may be given muscle relaxants to prevent movement during the shock. The defibrillator is placed in close proximity to the patient's chest and connected to the electrodes, which are then placed on the patient's chest. The defibrillator is activated by a medical professional, who delivers a controlled electrical shock to the patient's heart. Defibrillators are a critical tool in the treatment of cardiac arrhythmias and can help save lives in emergency situations. However, it's important to note that defibrillation is not a cure for arrhythmias and is typically used in conjunction with other forms of treatment, such as medication and lifestyle changes. In addition to their use in emergency situations, defibrillators are also used in medical research and training programs. Medical professionals are trained on how to use defibrillators safely and effectively, and many hospitals and clinics have protocols in place for the rapid deployment of defibrillators in emergency situations. Defibrillators are a critical tool in the treatment of cardiac arrhythmias and can help save lives in emergency situations. While they may seem intimidating, these devices are designed to be used by trained medical professionals and are an important part of modern healthcare. Sodium Sulfide is a yellow to brick-red crystalline solid that is soluble in water. Sodium Sulfide is widely used in various industries, including the chemical, textile, and paper industries. Sodium Sulfide is a highly reactive compound that reacts with water to produce hydrogen Sulfide, a highly toxic gas. It has a strong odor of rotten eggs and is highly corrosive. Sodium Sulfide is an alkali metal salt and is highly soluble in water, making it easy to handle and transport. Sodium Sulfide is primarily produced by the reduction of sulfur using sodium. The reaction occurs at high temperatures in a reducing atmosphere. The resulting sodium Sulfide is then purified by recrystallization. Sodium Sulfide can also be produced by the reaction of sodium hydroxide and sulfur under high pressure and temperature. Sodium Sulfide is widely used in the leather industry as a dehairing agent. It helps to remove hair and other unwanted materials from animal hides, making them suitable for use in leather products. It is used in the textile industry to remove impurities from fabrics and to dye them. It is also used as a reducing agent in the production of sulfur dyes. Sodium Sulfide is used in the textile industry to remove impurities from fabrics and dye them. It is also used as a reducing agent in the production of sulfur dyes. Sodium Sulfide can be added directly to the dye bath to help the dye penetrate the fabric and produce a deep, even color. Sodium Sulfide is used in the pulp and paper industry to dissolve lignin, a complex organic compound that binds fibers together. This makes it easier to separate the fibers and produce high-quality paper. It is used in the chemical industry as a reducing agent and as a source of Sulfide ions. It is also used in the production of sulfuric acid, which is used in a wide range of industrial processes. Sodium Sulfide is used in the mining industry to separate metal Sulfides from gangue minerals. It is also used to treat wastewater from mining operations. Sodium Sulfide is used in the chemical industry as a reducing agent and as a source of Sulfide ions. It is also used in the production of sulfuric acid, which is used in a wide range of industrial processes. Sodium Sulfide can be added directly to the reaction mixture to provide Sulfide ions or to reduce other chemicals. Sodium Sulfide is used in the mining industry to separate metal Sulfides from gangue minerals. It is also used to treat wastewater from mining operations. Sodium Sulfide is added to the ore during the flotation process to create a frothy mixture that separates the metal Sulfides from the gangue minerals. Sodium Sulfide is a highly reactive compound that can cause serious health and safety hazards if not handled properly. It is a strong reducing agent and reacts violently with oxidizing agents, producing hydrogen Sulfide gas. Sodium Sulfide is also highly corrosive and can cause severe burns and tissue damage upon contact with the skin. It is important to note that the specific application and concentration of sodium Sulfide will vary depending on the industry and intended use. It is recommended to follow manufacturer guidelines and safety regulations when using sodium Sulfide. When handling sodium Sulfide, it is important to wear protective clothing, gloves, and eyewear to prevent skin and eye contact. Sodium Sulfide should be stored in a cool, dry, well-ventilated area, away from oxidizing agents and sources of heat and ignition. In case of contact with the skin or eyes, it is important to flush the affected area with plenty of water and seek medical attention immediately. Ingestion of sodium Sulfide can be fatal, and emergency medical attention should be sought if ingested. Humic Acid is an organic compound that is naturally found in soil and other natural materials. It is a complex mixture of different organic molecules that are formed during the decomposition of plant and animal matter. Humic acid is an important component of soil, and it plays a critical role in soil fertility, plant growth, and nutrient cycling. Humic acid is composed of a variety of different organic molecules, including fulvic acid, humin, and humic substances. These molecules are formed through the breakdown of organic matter by microbes and other organisms in the soil. The breakdown of organic matter is a natural process that occurs when plants and animals die and decompose. During this process, the organic matter is broken down into its constituent parts, including humic acid. Humic Acid has a wide range of functions in soil. One of the most important functions is to improve soil fertility. Humic acid contains a range of nutrients, including nitrogen, phosphorus, and potassium, as well as trace elements such as calcium, magnesium, and iron. These nutrients are released slowly over time, providing a steady source of nutrition for plants. In addition to providing nutrients, humic acid also helps to improve soil structure. It has a high cation exchange capacity, which means that it can hold onto and release nutrients as needed. This helps to prevent nutrient leaching, which can occur when nutrients are washed out of the soil by rainfall or irrigation. Humic Acid also helps to improve soil water-holding capacity. It has a sponge-like structure that can hold onto water molecules, making it easier for plants to access water even during dry periods. This helps to reduce the need for irrigation and can help to conserve water resources. Humic acid has also been shown to have a range of other benefits for plants. It can help to improve root development, increase seed germination rates, and improve plant growth and yield. It has also been shown to increase plant resistance to stress, including drought, salinity, and high temperatures. Humic acid is often added to soil as a soil amendment. It can be added in the form of organic matter, such as compost or manure, or as a commercial product. Commercial humic acid products are typically derived from natural sources such as peat or lignite, and they are available in liquid, granular, or powder form. When using Humic Acid as a soil amendment, it is important to follow the recommended application rates. Applying too much humic acid can lead to nutrient imbalances or other problems in the soil. It is also important to choose a high-quality product that has been tested and proven to be effective. In addition to its use as a soil amendment, Humic Acid is also used in a range of other applications. It is used in the production of fertilizers, as a feed additive for livestock, and as a food supplement for humans. It has also been shown to have potential applications in the fields of medicine and biotechnology. Herbicides that are soil-active affect the roots, seeds, and shoots of the plant. Soil Active Herbicides are integrated into the soil and so aid in preventing the growth of weeds and undesirable plants. Despite its many benefits, there are some potential drawbacks to using humic acid. One of the main concerns is that it can be expensive, particularly when used on a large scale. It can also be difficult to apply effectively, particularly in areas with heavy clay soils or other soil types that are difficult to amend. Another potential concern is the impact of Humic Acid d on the environment. While it is a natural compound, there is some evidence to suggest that high levels of humic acid in soil can lead to the buildup of heavy metals or other contaminants. This can be a particular concern in areas with high levels of pollution or industrial activity. Humic acid is an important organic compound that plays a critical role in soil fertility, plant growth, and nutrient cycling. It is composed of a variety of different organic molecules. Dehydration Products Contain Sodium Metabisulfite To Replace Electrolytes And Treat Dehydration9/3/2023 An inorganic molecule with the chemical formula Na2S2O5 is Sodium Metabisulfite, also known as sodium pyrosulfite (IUPAC spelling; Br. E. sodium metabisulphite or sodium pyrosulphite). The compound is also known as disodium metabisulfite. It functions as a bactericide, antioxidant, and preservative. Sodium bisulfite is created when it dissolves in water. In order to stop the oxidation of the adrenaline in drugs containing epinephrine, Sodium Metabisulfite is added as an excipient. The formulation of epinephrine autoinjectors like the EpiPen and combination medication formulations including a local anaesthetic and adrenaline, for instance, both include it. Although adrenaline combines with the sodium metabisulphite, causing it to break down and produce epinephrine sulphonate, this extends the shelf life of the formulation. It is employed as a rust-stain remover in conjunction with sodium hydrosulfite. Photography use it. Tree stumps can be removed using concentrated sodium metabisulfite. Certain brands, which include 98% sodium metabisulfite, lead to the lignin in the stumps being broken down, making it easier to remove them. Several pills, including those containing paracetamol, also employ it as an excipient. This substance's ability to be added to a blood smear in a test for sickle cell anaemia is a highly significant part of its health-related properties (and other similar forms of haemoglobin mutation). By a complicated polymerization, the chemical makes dead cells sickle, indicating illness. In order to make coconut cream, it serves as a bleaching agent. It (or liquid SO2) is frequently employed as an antibacterial and antioxidant in the production of wine; in the US, bottled wine has the label "Contains Sulfites" to make this clear. Sodium Metabisulfite functions as a reducing agent to dissolve sulphide bonds in garments composed of natural fibres that have shrunk, enabling the garment to regain its former shape after washing. In industrial gold cyanidation procedures, it serves as a source of SO2 for the breakdown of cyanide when combined with air or oxygen. When combined with oxygen or air, it is utilised as a source of SO2 to precipitate elemental gold in chloroauric (aqua regia) solutions. It is applied to quench lingering chlorine in the water treatment sector. It is applied while etching samples of iron-based metal for microstructural examination. The third-lightest halogen compound and one with an atomic number of 35 is Bromine Derivatives. At room temperature, it is a fuming reddish-brown liquid with characteristics that are essentially identical to those of chlorine and iodine. When shipping consumer items like shoes and apparel, it is used as a fungicide to prevent mould and microbes. Prior to delivery, the anhydrous, sodium metabisulfite solid active component is put to plastic stickers and packaging (such Micro-PakTM). During transport, the devices take moisture from the air and emit small amounts of sulphur dioxide. Fruit is sent using it to keep it fresh. Sodium Metabisulfite serves as a solvent in the process of removing starch from cereal crops, fruits, and tubers. High pressure reverse osmosis and nanofiltration water desalination membranes are treated with it as a pickling agent for long-term storage in between usage. Although not being flammable, Sodium Metabisulfite breaks down at a temperature of 150 °C, generating hazardous fumes. As it dissolves in water, it becomes corrosive. It should be stored carefully since it is toxic if swallowed and corrosive to metals. Lithium Hydroxide is a compound composed of lithium, oxygen, and hydrogen, with the chemical formula LiOH. It is a white, odorless solid that is highly soluble in water, and is commonly used in a variety of applications such as batteries, ceramics, and as a reagent in organic chemistry. Lithium Hydroxide is produced by the reaction of lithium carbonate and water, resulting in the formation of lithium hydroxide monohydrate, which is then dried to form the anhydrous form of the compound. The resulting product is a fine powder that is stable at room temperature and pressure, and has a melting point of 462°C. One of the primary uses of Lithium Hydroxide is in the production of rechargeable lithium-ion batteries, which are used in a variety of electronic devices such as smartphones, laptops, and electric vehicles. Lithium hydroxide is a key component in the cathode of these batteries, which stores the electrical charge that powers the device. Lithium-ion batteries have become increasingly popular due to their high energy density and long cycle life, and lithium hydroxide is a crucial component in their manufacture. In addition to its use in batteries, Lithium Hydroxide is also used in the production of ceramics and glass, where it is added to glazes to improve their properties. It is particularly useful in the production of high-strength ceramics such as those used in aerospace applications, due to its ability to improve the strength and durability of the final product. Lithium hydroxide can also be used as a flux in glass production, where it lowers the melting point of the material and helps to improve its workability. Lithium hydroxide is also used as a reagent in organic chemistry, where it is used to convert carboxylic acids to lithium carboxylates, which can be used in a variety of reactions. It is particularly useful in the production of pharmaceuticals and agrochemicals, where it is used as a key intermediate in the synthesis of a wide range of compounds. In addition to its industrial uses, Lithium Hydroxide has also been investigated for its potential use in the treatment of certain medical conditions. Lithium is a well-known mood stabilizer, and lithium hydroxide has been studied for its potential use in the treatment of conditions such as bipolar disorder and depression. While lithium hydroxide is not used directly as a medication, it is a precursor to the lithium salts used in these applications. Despite its many uses, Lithium Hydroxide is not without its challenges. One of the primary issues is the limited availability of lithium, which is a relatively rare element in the earth's crust. This has led to concerns about the sustainability of lithium mining and production, particularly as demand for lithium-ion batteries continues to grow. In addition, the production of lithium hydroxide can be energy-intensive, requiring significant amounts of electricity to convert lithium carbonate to lithium hydroxide. An inorganic substance, Lithium Carbonate is the lithium salt of carbonate and has the formula Li2CO3. This white salt is frequently used to treat mood disorders and to process metal oxides. The Term Polyphenol Refers To Several Thousand Plant-Based Molecules That Are Antioxidants26/1/2023 Polyphenols are a diverse group of naturally occurring compounds found in plants, including fruits, vegetables, herbs, spices, and even beverages such as tea and wine. They are known for their antioxidant properties, which means they can neutralize harmful molecules called free radicals and protect cells from damage. There are many different types of polyphenols, including flavonoids, anthocyanins, catechins, and tannins. Each type of polyphenol has its own unique chemical structure and health benefits. For example, flavonoids are known for their anti-inflammatory properties, while anthocyanins are known for their ability to improve cardiovascular health. Research has also shown that Polyphenol may help to reduce the risk of certain types of cancer, particularly cancers of the digestive system. They may also help to improve brain function and reduce the risk of neurodegenerative diseases such as Alzheimer's and Parkinson's. Polyphenols are found in a wide variety of foods and beverages, with some of the richest sources being berries, dark chocolate, green tea, red wine, and extra-virgin olive oil. Eating a diet rich in fruits, vegetables, and whole grains is the best way to get a good intake of polyphenols. It's important to note that polyphenols can also be found in supplements, but it's best to get them through a healthy diet as taking supplements may not always be safe. In addition, polyphenols are heat-sensitive, meaning that cooking or processing can reduce their levels in food. The best way to preserve polyphenols in food is to eat it raw or to use gentle cooking methods such as steaming or sautéing. Overall, Polyphenols are a diverse group of compounds that have many health benefits, including antioxidant, anti-inflammatory, and anticancer properties. Incorporating foods rich in polyphenols into your diet is an easy and delicious way to support your overall health and well-being. In the natural world, polyphenols have a very specific role in protecting plants from pests and illnesses as well as providing them with a vibrant colour. The animals that consume the vegetables subsequently spread the seeds. It is an ingenious method of reproduction! Ironically, this protection system aids us as people. A family of water-soluble compounds called Polyphenol is widely present in plants. A number of phenolic groups that together form a more or less complicated structure with a typically high molecular weight define them. Plants utilise these phytonutrients as part of their defence mechanisms against intruders. If they come from many sources, humans can benefit greatly from them in a number of ways. In actuality, while some polyphenols, like quercetin, are prevalent in all plants, others can only be found in specific foods. Indole found in cabbage and anthocyanins found in several berries, drupes, and ancillary fruits fall within this category. Their protective function stops cell oxidation, which aids in the battle against cellular ageing. They are mostly recognised for their ability to prevent inflammatory, cardiovascular, neurodegenerative, and some forms of cancer. Food flavour and colour are also affected by Polyphenols! Among flavonoids, the flavonone sub-group, which gives grapefruits their bitter flavour, tannins, which give many fruits their astringency (such as the peel and seeds of grapes), and anthocyanins, which give red berries their colour, are a few examples. |
Categories
All
|