|
The field of medicine has witnessed remarkable advancements with the introduction of Biologics & Biosimilars. Biologics, derived from living organisms, and biosimilars, highly similar versions of biologic products, have revolutionized therapeutic solutions. This article explores the pioneering advances in biologics and biosimilars and their impact on the medical landscape. From their development and manufacturing to their therapeutic applications, we will delve into the scientific breakthroughs, regulatory considerations, and benefits these innovations offer in terms of patient care and outcomes. MENA Biologics & Biosimilars manufacturing is a complex process that demands stringent quality control measures. The production of biologics often involves genetically modified cell lines, such as mammalian cells or bacteria, which serve as factories for protein synthesis. Bioreactors and purification techniques are employed to obtain pure and high-quality biologic products. Manufacturing processes must adhere to strict regulations and guidelines to ensure consistent batch-to-batch quality, safety, and efficacy. Biologics are medicinal products derived from living organisms, such as proteins, antibodies, and nucleic acids. These complex molecules exhibit high specificity and efficacy in targeting disease pathways. Unlike traditional small-molecule drugs, biologics are manufactured through biotechnological processes that involve recombinant DNA technology, cell culture systems, and protein purification techniques. The precise nature of biologics enables them to interact with specific cellular targets, leading to more targeted and personalized treatment approaches. Biosimilars, on the other hand, are highly similar versions of approved biologic products. They undergo rigorous comparison studies to demonstrate similarity in terms of quality, efficacy, and safety to their reference biologic. While biosimilars offer cost-effective alternatives, their development requires extensive analytical and clinical studies to ensure similarity to the reference product. Regulatory bodies have established guidelines and frameworks to govern the approval process for biosimilars, ensuring their safety and efficacy. The development of biologics involves a multidisciplinary approach, encompassing various stages from target identification to clinical trials. Advances in genetic engineering, protein expression systems, and cell culture techniques have accelerated the production of biologics. Scientists employ state-of-the-art technologies to modify and optimize therapeutic proteins, enhancing their stability, efficacy, and bioavailability. This enables the development of novel biologics with improved pharmacokinetic and pharmacodynamic properties. Biologics & Biosimilars have found extensive applications in various therapeutic areas, including oncology, autoimmune diseases, and infectious diseases. Monoclonal antibodies, a prominent class of biologics, have transformed cancer treatment by targeting specific molecules involved in tumor growth. Biosimilars have also played a significant role in expanding patient access to life-saving treatments by offering cost-effective alternatives to expensive biologic therapies. The hereditary condition known as Cystic Fibrosis (CF) typically affects the lungs but can also have an impact on the pancreas, kidneys, liver, and gut. Recurrent lung infections can have long-term effects for Cystic Fibrosis patients, including breathing problems and coughing. Other symptoms and indicators include guild of the fingers and toes, poor growth, male infertility, fatty stool, and sinus infections. Different people may have symptoms in varying degrees. The approval process for Biologics & Biosimilars involves rigorous assessment by regulatory authorities, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Regulatory agencies evaluate the quality, safety, and efficacy of these products based on comprehensive data obtained from analytical and clinical studies. Pharmacovigilance programs continuously monitor the safety profiles of biologics and biosimilars post-approval, ensuring ongoing assessment of their benefit-risk balance. Biologics And Biosimilars have emerged as pioneering therapeutic solutions, offering new avenues for personalized medicine and improved patient care. These advancements have transformed the treatment landscape across various medical fields, providing targeted therapies for complex diseases. Through scientific innovation, meticulous manufacturing processes, and stringent regulatory oversight, biologics and biosimilars continue to shape the future of medicine. As research and development efforts progress, it is anticipated that these groundbreaking therapies will further expand, positively impacting the lives of patients worldwide.
0 Comments
Next-generation DNA Sequencing technology has revolutionized the field of genomics, enabling researchers to delve deeper into the intricacies of the genetic code with unprecedented speed, accuracy, and cost-effectiveness. This breakthrough has had a profound impact on various areas of scientific research, clinical diagnostics, and personalized medicine. In this essay, we will explore the remarkable impact of next-generation DNA sequencing technology and its implications for advancing our understanding of genetics and improving human health. Next-generation DNA Sequencing, also known as high-throughput sequencing, refers to a set of modern sequencing techniques that allow rapid sequencing of large volumes of DNA in parallel. Unlike traditional Sanger sequencing, which relied on capillary electrophoresis and fluorescent labeling, next-generation sequencing methods employ massively parallel sequencing platforms, allowing the simultaneous analysis of millions of DNA fragments. One of the significant impacts of next-generation DNA Sequencing technology lies in its ability to generate vast amounts of genetic data. This has facilitated the completion of large-scale genomic projects, such as the Human Genome Project, with remarkable efficiency. The increased speed and throughput of next-generation sequencing platforms have accelerated the pace of genomic research, enabling scientists to unravel the complexities of the human genome and gain insights into the underlying genetic basis of diseases. Next-generation DNA sequencing has revolutionized the field of cancer genomics. By sequencing the genomes of tumor cells, researchers can identify specific mutations and genetic alterations that drive the development and progression of various types of cancer. This knowledge has paved the way for personalized cancer treatments, as clinicians can now select targeted therapies based on the unique genetic profile of each patient's tumor. Additionally, next-generation sequencing has contributed to the discovery of novel cancer biomarkers, aiding in early detection and prognosis assessment. The impact of next-generation DNA Sequencing technology extends beyond cancer research. In the field of infectious diseases, sequencing the genomes of pathogens has provided valuable insights into their evolution, transmission patterns, and drug resistance mechanisms. This information is crucial for designing effective strategies to combat infectious diseases, develop vaccines, and track the spread of outbreaks. Furthermore, next-generation DNA sequencing has revolutionized the field of genetic research, allowing scientists to investigate the role of genetic variations in human health and disease. By sequencing the genomes of large populations, researchers can identify genetic variants associated with various conditions, including complex diseases like diabetes, cardiovascular disorders, and psychiatric illnesses. This knowledge opens up new avenues for developing personalized therapies, predicting disease risks, and tailoring treatment strategies based on an individual's genetic makeup. In addition to its impact on research, next-generation DNA Sequencing technology has transformed clinical diagnostics. The ability to rapidly and comprehensively sequence an individual's genome or exome (the protein-coding region of the genome) has enabled more accurate and efficient diagnosis of rare genetic disorders. Clinicians can now identify disease-causing mutations, even in cases where the clinical presentation is atypical or ambiguous. Early diagnosis through genomic sequencing allows for timely interventions, personalized management plans, and informed family counseling. The accurate order of the nucleotides within a DNA molecule can be determined using DNA Sequencing Equipment And Services, sometimes referred to as a DNA sequencer. Technically referred to as being read, the instrument reports the text string in terms of the four bases guanine (G), cytosine (C), adenine (A), and thymine (T). As the machine is able to analyze the light signals originating from fluorochromes bound to nucleotides, DNA sequencers also serve as optical instruments. The impact of next-generation DNA sequencing is not limited to human genomics. It has revolutionized fields such as agriculture, evolutionary biology, and conservation genetics. Sequencing the genomes of economically important crops and livestock has facilitated the identification of beneficial traits and the development of improved breeding strategies. In evolutionary biology, next-generation sequencing has shed light on the processes driving adaptation, speciation, and the preservation of biodiversity. Conservation geneticists use DNA Sequencing to study endangered species, assess population health, and guide conservation efforts. Stem Cell Manufacturing has emerged as a revolutionary field in healthcare, holding immense promise for transforming the way we treat diseases and injuries. By harnessing the remarkable potential of stem cells, scientists and researchers are developing innovative therapies and regenerative medicine solutions. This article explores the role of stem cell manufacturing in healthcare, discussing its principles, applications, challenges, and future prospects. Stem Cell Manufacturing involves the controlled and scalable production of stem cells in laboratory settings. Stem cells are undifferentiated cells capable of self-renewal and differentiation into specialized cell types. These unique properties make them valuable for repairing damaged tissues, regenerating organs, and developing personalized treatments. Stem cell manufacturing utilizes various techniques, such as cell culture, bioreactors, and tissue engineering, to grow and expand stem cells in a controlled environment. Stem cell manufacturing has tremendous potential for treating a wide range of diseases and conditions. For example, in the field of cardiology, stem cells can be used to regenerate heart muscle after a heart attack. In neurology, they hold promise for repairing damaged neural tissue in conditions like Parkinson's and spinal cord injuries. Stem cell therapies have also shown potential in orthopedics, ophthalmology, diabetes treatment, and autoimmune disorders. By manufacturing stem cells in large quantities, researchers can overcome limitations in supply and develop scalable therapies. Significant advancements have been made in stem cell manufacturing techniques. Traditional methods relied on the isolation of stem cells from embryonic sources, which posed ethical concerns and limited their availability. However, the development of induced pluripotent stem cells (iPSCs) revolutionized the field. iPSCs are adult cells reprogrammed to possess embryonic stem cell-like properties. They can be derived from a patient's own cells, eliminating the risk of immune rejection and ethical dilemmas. Moreover, new technologies like gene editing and 3D bioprinting have enhanced the precision and efficiency of stem cell manufacturing, opening up new possibilities for tailored therapies. While the potential of stem cell manufacturing is exciting, several challenges and safety considerations must be addressed. One key challenge is ensuring the quality and consistency of manufactured stem cells. Standardization of manufacturing processes and strict quality control measures are crucial to maintain reproducibility and safety. Additionally, the risk of tumorigenicity and uncontrolled cell growth needs to be carefully managed to avoid potential complications. Regulatory frameworks play a vital role in ensuring the safety and efficacy of stem cell-based therapies, and collaboration between regulatory bodies, researchers, and manufacturers is essential. The commercialization of stem cell manufacturing has gained momentum in recent years. Several companies have emerged, specializing in the production and distribution of stem cell-based therapies. This evolving market landscape presents opportunities for innovation, investment, and strategic partnerships. However, it also requires navigating complex regulatory pathways, addressing reimbursement challenges, and demonstrating clinical efficacy and cost-effectiveness. The future of stem cell manufacturing holds great promise. Ongoing research aims to improve the efficiency of stem cell production, develop novel differentiation protocols, and enhance the scalability of manufacturing processes. With advancements in bioprocessing techniques and automation, the cost of production is expected to decrease, making stem cell therapies more accessible. Additionally, the integration of stem cell manufacturing with other emerging technologies, such as gene editing and artificial intelligence, could lead to further breakthroughs and personalized treatments. The demand for Biotechnology Instrument is rising quickly along with the rise of chronic disease (which causes an increase in surgical procedures). The World Health Organization (WHO) estimates that, each year, 17.9 million people die from NCDs, with diabetes accounting for 2.0 million of those deaths (including kidney disease deaths brought on by diabetes), followed by cancer (9.3 million), chronic respiratory disease (4.1 million), and cancer (9.3 million). Stem cell manufacturing is revolutionizing healthcare by unlocking the potential of stem cells for disease treatment and regenerative medicine. Through controlled and scalable production, stem cell manufacturing enables the development of innovative therapies with the potential to transform the lives of millions. Despite challenges in quality control, safety, and regulatory compliance, the field continues to progress rapidly, driven by advancements in techniques, commercialization efforts, and supportive regulatory frameworks. As we move forward, collaborations between researchers, manufacturers, clinicians, and regulatory bodies will be crucial in realizing the full potential of stem cell manufacturing and delivering safe, effective, and personalized healthcare solutions to patients worldwide. Read The Press Release Of Stem Cell Manufacturing Hepatitis is a viral infection that affects the liver and can lead to serious health complications. To prevent the spread of hepatitis and protect individuals from its potential risks, vaccines have been developed for various types of hepatitis viruses. Hepatitis is an inflammation of the liver caused by hepatitis viruses, including hepatitis A, B, C, D, and E. Each type of virus differs in transmission routes, severity, and long-term effects. Hepatitis A and E are primarily spread through contaminated food and water, while hepatitis B, C, and D are transmitted through blood, sexual contact, and other body fluids. Types of Hepatitis Vaccines: a. Hepatitis A Vaccine: The hepatitis A vaccine provides protection against hepatitis A virus (HAV). It is recommended for individuals at risk of infection or those who wish to be immunized. The vaccine consists of two doses administered six to 12 months apart. It is safe and highly effective in preventing HAV infection. b. Hepatitis B Vaccine: The hepatitis B vaccine protects against the hepatitis B virus (HBV). It is recommended for all infants at birth and for individuals at risk of infection, including healthcare workers, people with multiple sexual partners, and injection drug users. The Hepatitis Vaccine is administered in a series of three to four doses over six months. It is highly effective in preventing chronic HBV infection and its complications. c. Combination Hepatitis A and B Vaccine: For individuals who require protection against both hepatitis A and B viruses, a combination vaccine is available. This vaccine simplifies the immunization process by providing simultaneous protection against both viruses. The doses and schedule may vary depending on the specific vaccine formulation. d. Hepatitis E Vaccine: Although less common, hepatitis E can cause severe illness, particularly in pregnant women. A vaccine for hepatitis E is available in some countries, but its availability may vary. It is recommended for individuals living in regions with a high prevalence of hepatitis E or those at increased risk of exposure. The immunization schedules for hepatitis vaccines vary based on the type of vaccine and individual characteristics. Here are the general recommendations: Hepatitis A Vaccine: Children: Two doses between 12 and 24 months of age. Adolescents and Adults: Two doses given at least six months apart. Hepatitis B Vaccine: Infants: A series of three doses: at birth, one to two months, and six months. Children and Adolescents: Three doses given over a six-month period. Adults: Three or four doses depending on the vaccine formulation. Vaccines support a healthy immune system and guard against deadly illnesses including hepatitis, polio, tuberculosis, and others. Immunisations aid the body's defences against diseases including bacteria and viruses. The body is only exposed to a very small yet safe quantity of weak or dead viruses or bacteria when receiving a Vaccines. The immune system is able to identify and combat infections if the body is exposed to them later in life. This is a natural way to treat contagious illnesses like pneumonia, measles, polio, and more. Combination Hepatitis A and B Vaccine: The dosing schedule may vary, but generally requires two to three doses.
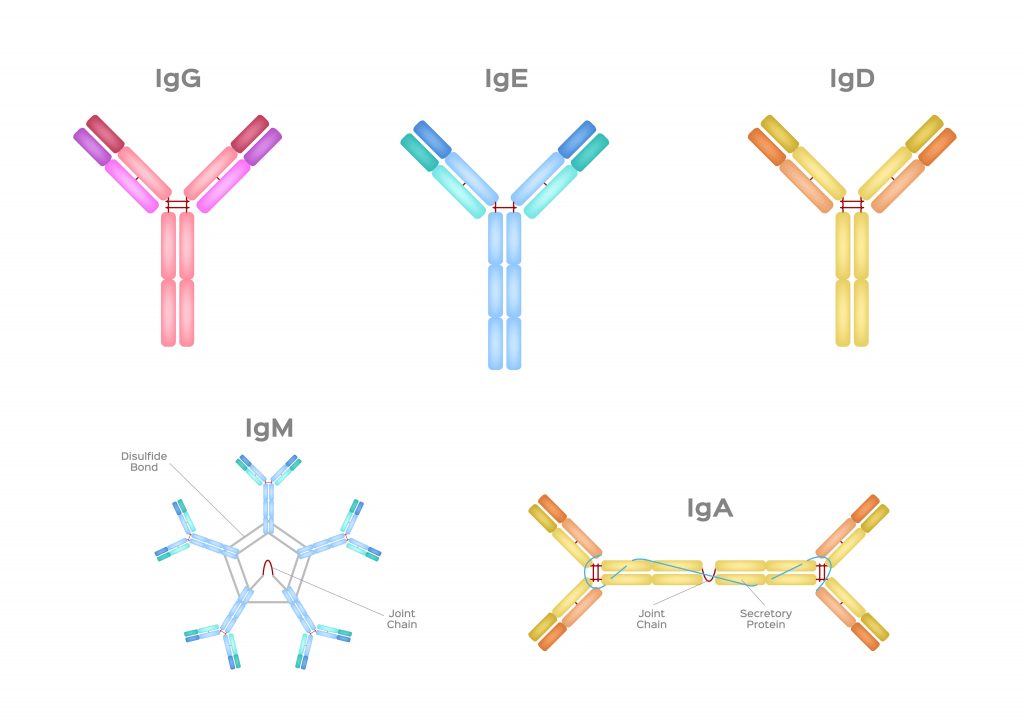
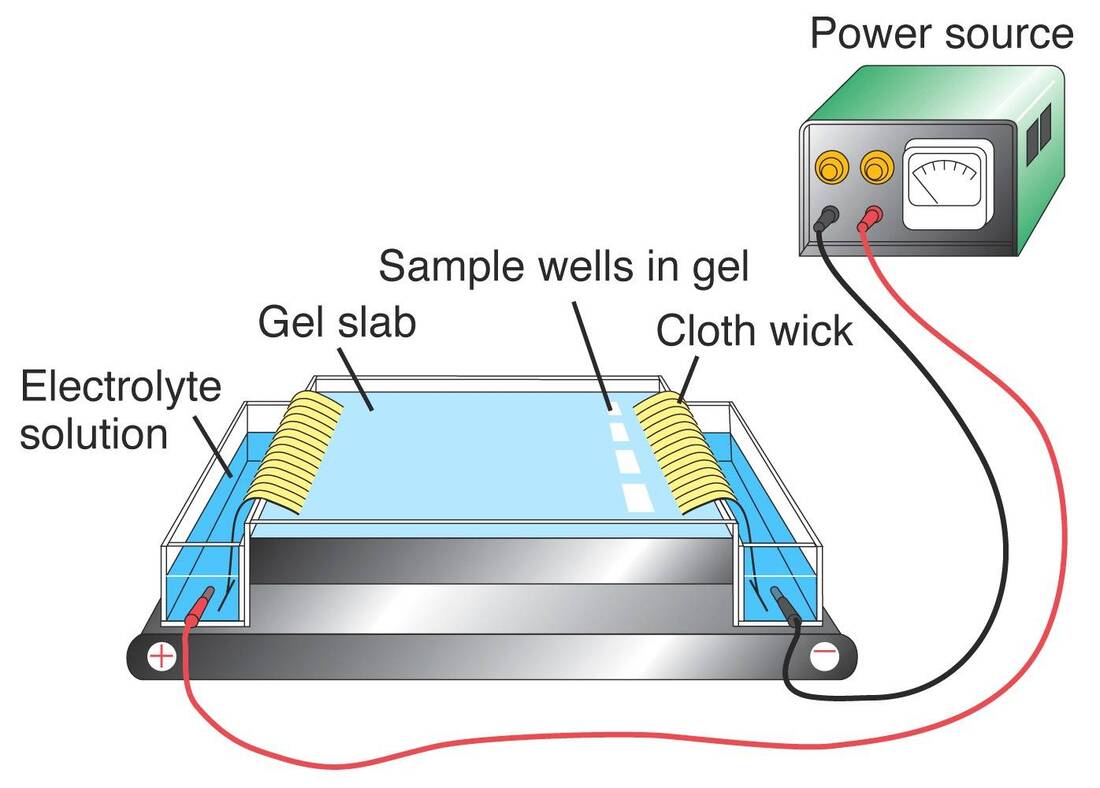
b. Post-Exposure Prophylaxis: For individuals who have been exposed to hepatitis A or B virus, specific post-exposure prophylaxis measures, including vaccination, may be recommended to prevent infection. c. Vaccine Safety: Hepatitis vaccines are generally safe, with the most common side effects being mild and transient, such as soreness at the injection site or low-grade fever. Severe adverse reactions are rare. d. Vaccination during Pregnancy: Hepatitis A and B vaccines are considered safe for pregnant women. However, the decision to vaccinate should be made in consultation with a healthcare provider. Immunoglobulins, also known as antibodies, are proteins that play a crucial role in the immune system's response to infections and diseases. They are produced by white blood cells known as B cells and are designed to recognize and neutralize specific pathogens or foreign substances that enter the body. Measuring immunoglobulin levels is an important tool in diagnosing a variety of diseases, as it can provide critical information about a patient's immune system health and function. There are five main classes of Immunoglobulin: IgA, IgD, IgE, IgG, and IgM. Each class has a unique structure and function, and measuring the levels of these classes can provide important insights into a patient's immune status. One of the most common uses of measuring immunoglobulin levels is in the diagnosis of primary immunodeficiency disorders (PIDs). PIDs are a group of inherited disorders that affect the immune system's ability to fight off infections. Individuals with PIDs may have recurrent infections, severe infections, or infections that are resistant to treatment. Measuring immunoglobulin levels can help identify PIDs by detecting deficiencies in the production of one or more classes of immunoglobulins. For example, patients with selective IgA deficiency have normal levels of other immunoglobulin classes, but low or absent levels of IgA. In addition to PIDs, measuring Immunoglobulin levels can also be useful in diagnosing autoimmune disorders. Autoimmune disorders occur when the immune system attacks the body's own cells and tissues. Measuring immunoglobulin levels can help identify the presence of autoantibodies, which are antibodies that target the body's own cells or tissues. For example, patients with rheumatoid arthritis may have high levels of rheumatoid factor, which is an autoantibody that targets joint tissues. Measuring immunoglobulin levels can also be used to monitor the progression of certain diseases. For example, in patients with multiple myeloma, a type of cancer that affects plasma cells, measuring immunoglobulin levels can help track the disease's progression and response to treatment. In multiple myeloma, cancerous plasma cells produce an excess of a single type of immunoglobulin, which can be detected in the blood. Measuring the levels of this immunoglobulin, known as a monoclonal protein or M protein, can help monitor the disease's progression and response to treatment. In addition to its diagnostic and monitoring applications, measuring Immunoglobulin levels can also be used to evaluate a patient's response to vaccination. Vaccines work by stimulating the immune system to produce antibodies against a specific pathogen. Measuring immunoglobulin levels after vaccination can help confirm that the patient has mounted an adequate immune response to the vaccine. For example, measuring IgG levels after vaccination for measles can confirm that the patient has developed immunity to the virus. Measuring immunoglobulin levels is an important tool in diagnosing and monitoring a variety of diseases. By providing critical information about a patient's immune system health and function, it can help guide treatment decisions and improve patient outcomes. Rabies is a viral disease that affects the nervous system and is transmitted to humans through the bite of an infected animal, most commonly dogs. The disease is fatal in nearly 100% of cases once symptoms appear, making prevention through vaccination crucial. The rabies vaccine was first developed by Louis Pasteur in 1885 and has since been refined and improved, with various types of vaccines now available. The most common type of vaccine is the inactivated rabies vaccine, which contains the killed rabies virus and is administered in a series of injections. The Rabies Vaccine is highly effective in preventing the disease when given before exposure and is recommended for individuals at high risk of exposure such as veterinarians, animal control personnel, and people who work with bats or other wildlife. It is also recommended for individuals traveling to areas where rabies is endemic or endemic with a high prevalence of rabies. The vaccination schedule typically involves an initial dose followed by one or more booster doses at regular intervals. The World Health Organization recommends a pre-exposure vaccination regimen of three doses of vaccine administered over four weeks. In the case of potential exposure to rabies, a post-exposure prophylaxis (PEP) regimen is recommended, which involves a series of injections of rabies vaccine and rabies immunoglobulin. The Rabies Vaccine is safe and well-tolerated, with most side effects being mild and short-lived. Common side effects include soreness or redness at the injection site, headache, and nausea. Serious allergic reactions are rare but can occur, and individuals with a history of severe allergic reactions should be closely monitored after vaccination. In addition to the traditional inactivated rabies vaccine, newer vaccines have been developed that offer more rapid and convenient protection. One such vaccine is the recombinant vaccine, which is made by inserting a gene from the rabies virus into a harmless virus or bacterium. This type of vaccine can be given in a single dose and protects within a week. Another newer vaccine is the oral Rabies Vaccine, which is used to control the spread of rabies in wildlife populations. The vaccine is placed in bait and distributed in areas where wildlife is known to carry rabies. When the animals eat the bait, they are immunized against rabies, helping to prevent the spread of the disease to other animals and humans. Despite the effectiveness of the rabies vaccine, rabies remains a significant public health concern in many parts of the world, particularly in developing countries where access to healthcare and veterinary services is limited. The World Health Organization estimates that rabies causes many deaths each year, with the majority of cases occurring in Asia and Africa. To address this issue, various organizations and governments have implemented programs to increase access to rabies vaccines and improve awareness of the importance of vaccination. These programs include mass vaccination campaigns for dogs and other animals, education campaigns for the public, and efforts to improve healthcare infrastructure in areas where rabies is endemic. Bioengineered (BE) for short, is the federal regulatory new word for GMOs. In the Bioengineered Food labeling law, specific Bioengineered foods comprising obvious modified genetic substances must reveal the occurrence of Bioengineered elements. The clause detectible modified genetic substances is important as it pretexts several goods that are prepared with GMOs from announcing the disclosure. Several items prepared with new GMO process such as RNAi, TALEN and CRISPR are currently untestable. Without a commercially accessible test, the modified genetic substance is unnoticeable and hence those foods would not need a Bioengineered label. In Addition, several processed foods consist vastly refined components made from GMOs. The process usually leaves no visible changed genetic substances behind in the end product, and hence those goods also will not need labels. Common household goods that consist components such as sugar prepared from GMO sugar beets or cooking oil prepared from GMO canola would fall into this category. Some food consisting noticeable modified genetic substances will need a Bioengineered Food disclosure. The USDA’s present List of Bioengineered Foods consist Canola, Ringspot virus-resistant Papaya, Alfalfa, Pink Pineapple, Summer squash, Corn, Soybean, Sugarbeet, Arctic™ Apple, AquAdvantage® Salmon, Potato, Cotton and Bt Eggplant. This USDA list shows which foods are referred to be bioengineered in their very basic, raw procedure. Anyhow, the way the Bioengineered law is written — with exceptions, loopholes and technical restrictions — several goods prepared from these bioengineered components will not need a revelation. The USDA states how this rule might affect labeling, utilizing a can of pork stew as an instance. A multi-compound canned stew might consist bioengineered components such as sweet corn. If pork is the major component listed first on the item panel, the item would not be focus to the Bioengineered labeling law. If the stew lists water, soup or stock as the first element and pork is known to be second on the element panel, the item would not need a Bioengineered label — even if the third element was GMO corn that is because water, stock and soup are ignored. Anyhow, if the stew consists more corn instead of pork, the item panel will list corn initially and a revelation would be required. The average shopper would be confused at this stage of complications. The USDA's instances determines the confusion built in the Bioengineered labeling law. A multi-component product might or might not be labeled Consisting Bioengineered Food Elements not based on the occurrence of bioengineered components however on the order in which the components are listed. Bioengineered is theoretical to mean GMO, however it utilizes a much more narrow description compared to users expect from other guarantees. According to the USDA’s definition, bioengineered foods must consist modified genetic substance which leaves out several goods made with GMOs. GMOs are utilized in around 80% of conventional administered foods in the US. From advocacy and teaching, the Non-GMO Project and collaborated companies have raised public awareness regarding GMOs in the food source. A 2020 survey from the Hartman Group reported that 97% of users were acquainted with the word GMO, than only 50% who stated acquaintance with bioengineering— showing a massive pool of individuals who do not know the term Bioengineered (BE) Food disclosure .Choosing the word bioengineered rather than the commonly utilized GMO shows transparency in the food system. Clarity, constancy and understanding are important tools in imparting data to the public. For instance, the meaning of bioengineered food depends upon specific language: consisting a necessity that Bioengineered Food must consist modified genetic substance and that the change is not otherwise available from traditional crossbreeding or identified in nature. In the meantime, the biotechnology sector is introducing new GMOs made with emerging methods — and they are usually arguing that those methods attain the similar outcomes as traditional crossbreeding, only faster. Yet, this dispute is reductive and deceptive — and it is extremely troubling while put to the definition of bioengineered foods. Rabies Vaccine is a syringe of two disarmed, cell-culture immunizers: HDCV or PCECV. The vaccine can be utilized in living beings to inhibit rabies post exposure or pre-exposure. Pre-exposure rabies vaccine is suggested for people in life-threatening jobs such as taxidermists, animal disorders control staff, veterinarians, wildlife ecologists and animal rehabilitators. It includes three syringes of a rabies vaccine one in the arm and booster dosage one in every two years, if the individual is found to be at risk. Post-exposure Rabies Vaccine is inoculated or injected in the shoulder muscle of elders and in the thigh of kids to those who have been in contact to a rabid animal. Prodromes of rabies take place within 3-10 weeks post being in contact and can scale from low to a severe, life-threatening disorder that comprises atypical behavior, illusions and hydrophobia. Once the prodromes take place, an individual is almost likely to die from rabies, until cured with a rabies vaccine. Earlier, less than 15 cases of unimmunized people living rabies once they have increased symptoms have been recognized. Rabies therapy comprise of a post-acquaintance immunization of an immunizer that is inoculated by syringe in the shoulder muscle and a plasma accessories known as HRIG. This immunizer and Rabies Immune Globulin are tough to detect so individuals may be fluttered to where it is accessible. The benefits of Rabies Vaccine are well renowned, and also curbs the spread of rabies to other individuals. While one get the rabies vaccination, the body creates antigens that defend it against this disorders. It is also protective for ladies who are gravid or breastfeeding, however the physician will require to advise the individual on this matter. Rabies is not a common disorder in U.S, however it can be deadly if one do not get clinical care instantly post being bitten or hurt by an animal. As per CDC, around 1 to 3 people suffer from rabies in a year in U.S. The movement of electrically charged particles in a fluid under the influence of an electric field is known as Electrophoresis, sometimes known as cataphoresis. The phenomena is known as electroosmosis if the liquid itself, rather than the particles, are what are set in motion, such as by a fixed diaphragm. Proteins and other colloids can be analysed and separated using electrophoresis, and it can also be used to cover objects like electron tube components with coatings. The use of electrophoresis as an analytical technique was first developed in 1930 by the Swedish scientist Arne Tiselius. The moving-boundary method of observation was developed by Tiselius, and it involves placing a layer of pure (i.e., particle-free) fluid over a quantity of the same fluid that contains colloidal particles. The boundary between the two layers of fluid is visible and moves at the rate of the particles' Electrophoresis. The mobility of ions in an electric field is the foundation of the separation method known as electrophoresis. Ions that are positively charged move towards a positive electrode whereas those that are negatively charged move towards a negative electrode. One electrode is typically at ground while the other is biassed positively or negatively for safety reasons. Ions can be separated because they migrate at varying rates based on their overall charge, size, and shape. The method is especially useful for macromolecules like proteins. These molecules are given a negative charge, which causes them to gravitate towards the positive electrode. A high-voltage supply, electrodes, a buffer, and a support for the buffer, such as filter paper, cellulose acetate strips, polyacrylamide gel, or a capillary tube, are all components of an electrode device. The other supports are typically employed for biological samples like protein mixes or DNA fragments, while open capillary tubes are used for a wide variety of samples. The support is stained to make it easier to see the separated components after a separation is finished. Isoelectric focusing has a significant positive impact on resolution. The support gel in this method maintains a pH gradient. When a protein descends the gel, it reaches the pH where its isoelectric point is located. The protein concentrates into a distinct band on the gel at this pH because it is neutral and no longer migrates. These are the Specific electrophoretic techniques:
The movement and separation of charged particles (ions) caused by an electric field is referred to as Electrophoresis. Two electrodes (anode, cathode) with opposing charges are joined by a conducting liquid known as an electrolyte to form an electrophoretic system. Differences in the ionic particle's velocity (v), which is the result of the particle's mobility (m) and the field strength (E), cause the separation effect: V= mE A particle's size, shape, charge, and separation temperature all affect an ionic particle's mobility (m), which is constant under specific electrophoretic circumstances. Electrical parameters (current, voltage, and power) as well as characteristics of the medium in which the particles are moving—such as ionic strength, pH level, viscosity, and pore size—are used to describe electrophoretic conditions. One of the main issues with the majority of Electrophoresis methods is the removal of heat produced by the flow of electric current. Every temperature difference alters the speeds of migration through the medium, distorting the bands of molecules that are separated from one another. It is obvious that doing electrophoretic studies at a constant temperature would be optimal. Pharmacogenomics is the study of genes and how medications alter a person's reaction. Pharmacogenomics is a developing field of science that combines pharmacology, the study of pharmaceuticals, with genomics, the study of genes, to provide safe, effective dosages of medication that are matched to the unique genetic makeup of each patient. One of the key programmes in which scientists are discovering and learning how genes relate to the way the body reacts to drugs is the Human Genome Project. In the future, it will be possible to forecast a person's medicine effectiveness based on genetic makeup and investigate the existence of adverse drug reactions. Pharmacogenomics is still in its infancy despite advances in science and technology. Pharmacogenomics is only occasionally used, but new methods are continually being tested in clinical settings. Pharmacogenomics will soon make it possible to create treatments that are specifically tailored to treat conditions like neurological, cardiovascular, HIV, cancer, and asthma. Pharmacogenomics examines how a patient's genes may affect how they react to medications. Medicines can operate better or worse depending on genetic variations in a patient. Also, they can aid in identifying which patients will experience adverse effects, ranging from the merely uncomfortable to potentially fatal. Pharmacogenomics can assist doctors in determining the dosage and type of medications to administer to patients. In the overall population, a medicine may carry a minimal risk of side effects, but one group that carries a particular allele may carry a high risk (a variation in their genome). Abacavir, an antiviral drug used in HIV combination therapy, serves as an illustration of this. The majority of individuals tolerate abacavir well, but a small percentage (about 5%) can experience a hypersensitivity reaction, which can be serious and occasionally deadly. According to NICE guidelines, patients should be screened for the gene variant HLA-B*5701 before starting treatment since it significantly increases their likelihood of experiencing a hypersensitive reaction. Researchers have discovered that this test, which has been available for more than ten years, significantly affects the frequency of hypersensitive reactions. A patient may metabolise a medicine more quickly than usual, which causes it to leave the body more quickly and not have the desired effect. Or a drug may be metabolised slowly by a person, accumulating in their system to the point where it can be harmful. Thiopurines, a class of medications with uses in both chemotherapy and immunosuppression for autoimmune illnesses, serve as an illustration of this. They may have harmful negative effects if used in excessive doses. In addition to the risks they provide, they may necessitate pausing therapy, which could lower the likelihood that the chemotherapy will be effective. Thiopurines are processed by an enzyme called Thiopurine Methyltransferase (TPMT). A working copy of the TMTP gene is absent in about 3 out of every 1,000 individuals, and closer to 10% have a less active version of the gene than the majority of the population. Some patients might become quite unwell if they receive a typical course of treatment. NICE does not currently recommend a gene test, but encourages doctors to "consider evaluating TPMT activity before initiating azathioprine, mercaptopurine, or tioguanine therapy. Individuals without TPMT activity should not be treated with thiopurine medications; those with low TPMT activity can be treated with care. Clinicians caring for leukaemia patients should eventually have access to whole genome sequencing as it becomes available to patients with difficult-to-treat malignancies and seriously unwell youngsters. |
Categories
All
|