|
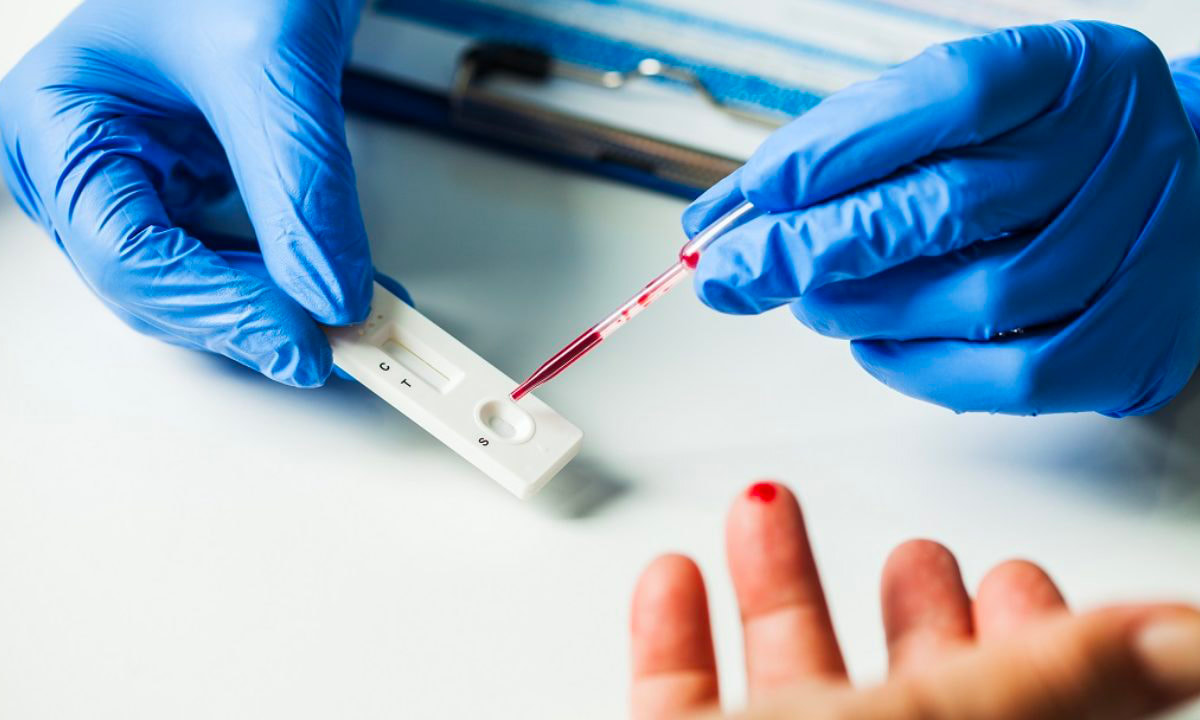
In the ever-evolving field of healthcare, one aspect that holds immense importance is diagnostics. The ability to accurately and swiftly diagnose diseases and conditions is crucial for effective treatment and patient care. Traditional diagnostic methods often involve time-consuming processes, leading to delays in diagnosis and treatment initiation. However, with the advent of rapid diagnostics, a paradigm shift has occurred, unlocking new levels of efficiency and transforming the healthcare landscape. Rapid Diagnostics refer to innovative techniques and technologies that allow for quick and accurate detection of diseases and conditions. These methods utilize advanced tools such as molecular diagnostics, point-of-care testing, biosensors, and microfluidics. Unlike traditional laboratory-based tests, rapid diagnostics provide results within minutes to hours, enabling healthcare providers to make timely decisions and initiate appropriate treatments. One of the primary benefits of Rapid Diagnostics is the significant reduction in turnaround time for test results. Traditional diagnostic methods often require samples to be sent to centralized laboratories, leading to prolonged waiting periods. In contrast, Rapid Diagnostics can be performed at the point of care, eliminating the need for sample transportation and enabling healthcare providers to make immediate decisions based on the results. By accelerating diagnoses, Rapid Diagnostics have a profound impact on patient care. In critical situations, such as emergency departments or intensive care units, quick access to test results can be life-saving. Rapid Diagnostics enable healthcare providers to promptly diagnose conditions such as heart attacks, sepsis, or infectious diseases, allowing for rapid intervention and improved patient outcomes. Rapid Diagnostics play a crucial role in advancing the field of precision medicine. With the ability to rapidly identify specific biomarkers, genetic mutations, or drug resistance patterns, healthcare providers can tailor treatments to individual patients. This personalized approach maximizes treatment effectiveness, minimizes adverse reactions, and optimizes resource utilization, thereby enhancing overall healthcare efficiency. The integration of Rapid Diagnostics into healthcare systems brings about streamlined processes and improved resource management. By minimizing the need for redundant tests and unnecessary procedures, Rapid Diagnostics reduce healthcare costs and alleviate the burden on healthcare infrastructure. Additionally, the ability to perform tests at the point of care reduces the need for patient referrals, enhancing convenience and accessibility, particularly in underserved areas. In the face of global health crises, Rapid Diagnostics emerge as powerful tools for infectious disease control. Rapid tests for pathogens, such as viruses or bacteria, enable early detection, containment, and prevention of outbreaks. By swiftly identifying infected individuals, appropriate measures can be implemented to limit transmission, safeguard public health, and prevent widespread epidemics. Testing samples at the point of care is known as point of care diagnostics, and it enables quick diagnosis and treatment planning. Early disease detection is made possible by point-of-care diagnostics, which also has a significant impact on how well a condition is treated medically. Point-Of-Care Diagnostics utilize lateral flow, agglutination tests, biosensors, and solid phase technologies, all of which are cutting-edge and specialized to certain situations. For instance, lateral flow technology is employed in several applications, including pregnancy testing. Rapid Diagnostics hold immense potential for remote and resource-limited settings, where access to healthcare facilities and laboratory infrastructure is limited. Portable and user-friendly rapid diagnostic devices enable healthcare providers in these areas to perform essential tests on-site, reducing the need for sample transportation and long waiting times. This empowers frontline healthcare workers, improves patient care, and contributes to health equity. The advent of Rapid Diagnostics has fueled research and development efforts in the field. As technology continues to advance, new diagnostic platforms, biosensors, and molecular assays are being developed, leading to further improvements in speed, accuracy, and affordability. These advancements not only enhance diagnostic capabilities but also open doors for innovative applications in areas such as monitoring chronic diseases, detecting cancer biomarkers, and predicting treatment response.
0 Comments
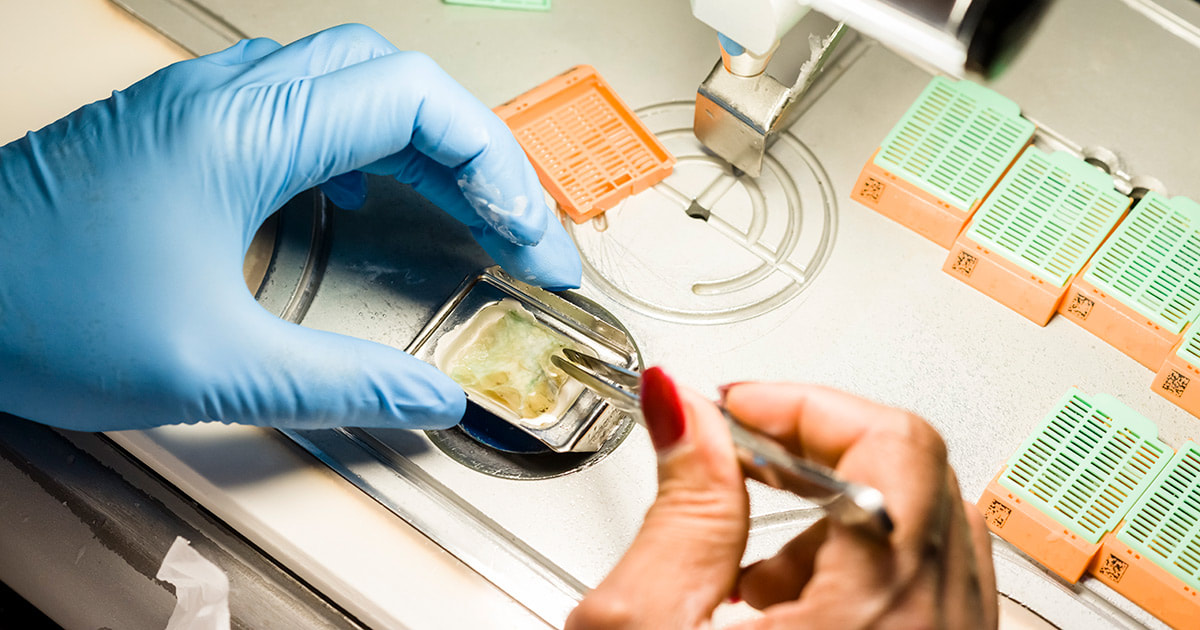
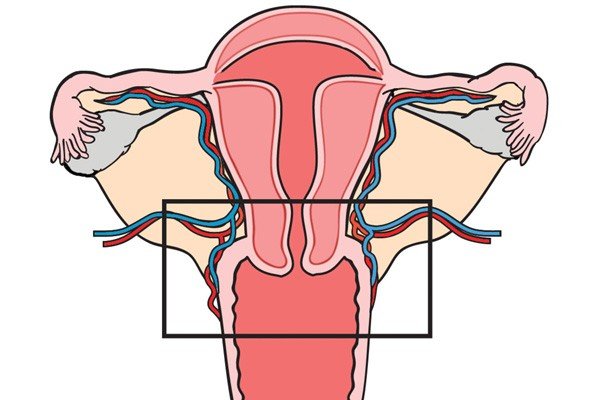
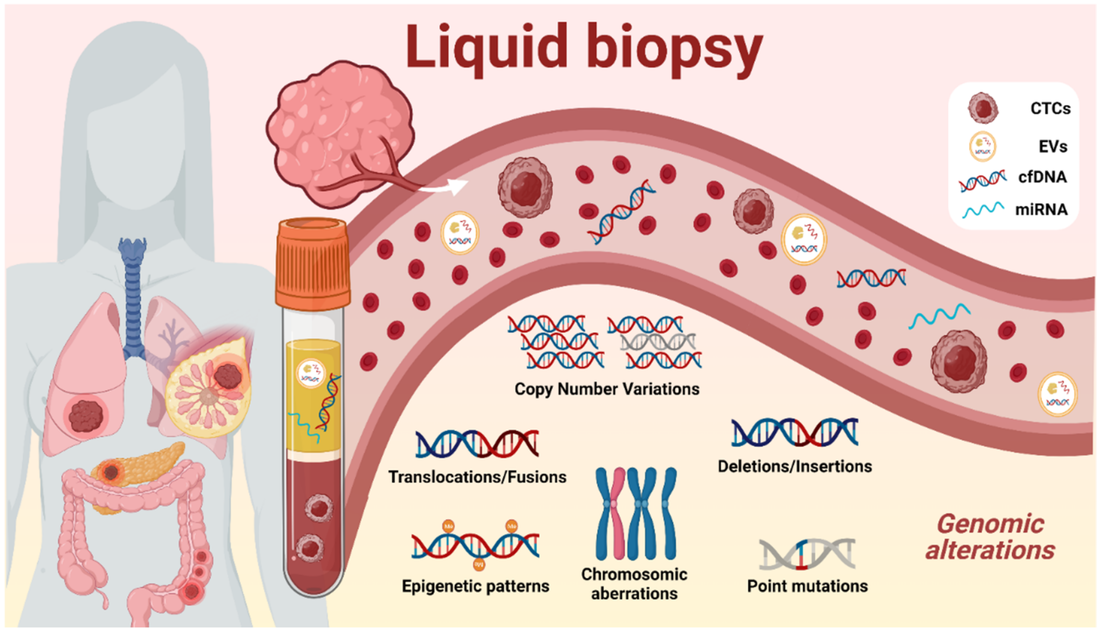
Anatomic Pathology serves as a crucial field in medicine, delving deep into the intricate world of tissues and organs to unravel the mysteries of disease. Like skilled artists, anatomic pathologists utilize a diagnostic canvas to create masterpieces of understanding, utilizing their expertise to interpret the microscopic landscape of tissues. At its core, anatomic pathology focuses on the examination of tissue specimens obtained through biopsies, surgeries, or autopsies. By employing a variety of techniques such as histology, immunohistochemistry, and molecular diagnostics, anatomic pathologists meticulously scrutinize these specimens to identify abnormal cellular and tissue changes indicative of disease. The analogy of a diagnostic canvas illustrates the intricate nature of Anatomic Pathology. Tissues become the medium, and pathologists become the artists, meticulously studying each brushstroke or cellular structure to create a comprehensive and accurate diagnostic interpretation. The canvas is the histological slide—a microscopic snapshot of the tissue's intricate details that holds the key to unlocking the underlying pathology. Every stroke on the diagnostic canvas tells a story. The pathologist's expertise enables them to identify and differentiate various cell types, recognize abnormal growth patterns, and detect alterations that point to specific diseases. This comprehensive analysis not only aids in establishing an accurate diagnosis but also provides critical information for treatment planning, prognosis assessment, and monitoring disease progression. Anatomic Pathology masterpieces extend beyond diagnosing common diseases. They also play a vital role in identifying rare and challenging conditions, uncovering the underlying causes of genetic disorders, and guiding precision medicine approaches. Through immunohistochemistry, pathologists can detect specific proteins expressed by cells, allowing for targeted therapies tailored to an individual's unique disease characteristics. Furthermore, the advent of molecular diagnostics has revolutionized anatomic pathology, providing deeper insights into diseases at the genetic level. Techniques like fluorescence in situ hybridization (FISH) and polymerase chain reaction (PCR) enable the detection of genetic alterations and mutations, aiding in the classification of tumors, prediction of therapeutic response, and identification of potential therapeutic targets. Anatomic pathology's impact extends to the field of research as well. Pathologists actively contribute to scientific advancements by collaborating with clinicians, molecular biologists, and other researchers. Their expertise in interpreting tissue morphology, coupled with molecular and genomic data, offers a comprehensive understanding of disease mechanisms and facilitates the development of new diagnostic tools and therapeutic interventions. In the realm of patient care, anatomic pathology forms a cornerstone in multidisciplinary teams. By providing precise and accurate diagnoses, pathologists contribute to treatment decisions, guide surgical interventions, and monitor treatment responses. They work closely with surgeons, oncologists, radiologists, and other healthcare professionals to ensure comprehensive care and optimize patient outcomes. Quality assurance is vital in anatomic pathology, ensuring that the diagnostic canvas remains consistent and reliable. Pathology laboratories undergo rigorous accreditation processes, implement stringent quality control measures, and participate in external proficiency testing programs to maintain the highest standards of accuracy and precision. These measures foster confidence in the diagnostic interpretations, enabling physicians to make informed clinical decisions. As technology continues to advance, anatomic pathology stands at the forefront of innovation. Digital pathology, for instance, enables the scanning and digitization of histological slides, facilitating remote consultations, inter-laboratory collaborations, and artificial intelligence-driven image analysis. These advancements enhance efficiency, reduce turnaround times, and promote standardized diagnoses. Anatomic Pathology represents a remarkable blend of artistry, scientific expertise, and clinical acumen. Through the diagnostic canvas, anatomic pathologists unravel the complexities of disease, creating masterpieces of understanding that guide clinical decisions, advance research, and improve patient outcomes. Chromatography Is A Technique For Separating Components Of A Mixture Based On Their Relative Amounts7/6/2023 Chromatography is a powerful analytical technique that plays a crucial role in various scientific fields, including chemistry, biochemistry, pharmaceuticals, environmental science, and forensic science. It is a separation method that enables scientists to unravel the complex mixture of compounds present in a sample, thereby revealing the secrets of its chemical composition. According To Coherent Market Insights, The Global Chromatography Market Was Valued At US$ 9,701.6 Mn In 2019 And Is Forecast To Reach A Value Of US$ 13,465.2 Mn By 2027 At A CAGR Of 4.8% Between 2020 And 2027. At its core, Chromatography is based on the principle of differential partitioning, where a mixture is separated into its individual components due to differences in their distribution between two phases: a stationary phase and a mobile phase. The stationary phase can be a solid or a liquid supported on a solid, while the mobile phase is typically a liquid or a gas. As the mobile phase moves through the stationary phase, different compounds interact differently and separate based on their affinity for each phase. This differential migration allows for the separation and identification of individual components in the mixture. Types of Chromatography: There are several types of Chromatography techniques, each offering unique advantages and applications. Some commonly used techniques include: 1. Gas Chromatography (GC): In GC, the mobile phase is a gas, and the separation is based on the interaction between the sample components and the stationary phase within a column. It is particularly suitable for volatile and semi-volatile compounds. 2. Liquid Chromatography (LC): LC involves the use of a liquid mobile phase and a stationary phase, which can be a solid (as in solid-phase extraction) or a liquid (as in high-performance liquid chromatography, HPLC). LC is versatile and widely used in various fields due to its ability to separate a wide range of compounds. 3. Thin-Layer Chromatography (TLC): TLC utilizes a thin layer of stationary phase, such as silica gel or cellulose, on a solid support. It is a simple and cost-effective technique, often used for qualitative analysis and compound identification. 4. High-Performance Thin-Layer Chromatography (HPTLC): HPTLC is an advanced version of TLC that provides higher resolution and sensitivity, making it suitable for more complex separations. Chromatography is a fundamental tool in unraveling the chemical composition of complex mixtures. By separating a mixture into its individual components, chromatography enables scientists to identify and quantify each compound present. This knowledge is vital in understanding the properties, behavior, and interactions of substances in various contexts. The combination of biology and information technology is referred to as bioinformatics. Bioinformatics use computer software tools for database building, maintenance, and creation. Additionally, it is utilised in data mining, communication networking, and data warehousing. Additionally, it is commonly used for managing biological data in the context of drug development and medical research. The history of Chromatography can be traced back to 1850 when a Russian botanist named Mikhail Tsvet first observed the separation of plant pigments on a solid adsorbent column. He coined the term "chromatography" from the Greek words "chroma" (color) and "graphein" (to write) to describe this phenomenon. However, it was not until several decades later, in the early 20th century, that chromatography began to gain recognition as a scientific technique. Metabolomics, a rapidly evolving field of study, focuses on the comprehensive analysis of small molecules known as metabolites within a biological system. It provides valuable insights into the biochemical pathways, cellular processes, and overall metabolic profile of an organism. To delve deeper into the complex world of metabolomics, advanced analysis services have emerged, offering researchers powerful tools and techniques to unlock the mysteries of metabolism. Metabolomics Services plays a crucial role in bridging the gap between genotype and phenotype, enabling a deeper understanding of how genes and environmental factors contribute to an organism's overall metabolic state. While basic metabolomics techniques provide a broad overview, advanced analysis services offer enhanced resolution, sensitivity, and specificity. By employing cutting-edge technologies and methodologies, these services facilitate the identification, quantification, and characterization of a wide range of metabolites, even those present in low abundance. This comprehensive approach opens up new avenues for studying metabolic pathways, biomarker discovery, drug development, and personalized medicine. Mass spectrometry lies at the heart of advanced metabolomics analysis. It enables the precise measurement of metabolite masses and their fragmentation patterns, allowing for identification and quantification. Techniques such as liquid chromatography-mass spectrometry (LC-MS) and gas chromatography-mass spectrometry (GC-MS) offer high sensitivity and specificity, enabling the detection of thousands of metabolites in a single analysis. NMR spectroscopy provides valuable structural information about metabolites. It offers a non-destructive, quantitative, and reproducible analysis of complex mixtures. NMR-based Metabolomics Services complement mass spectrometry, enhancing the overall coverage and confidence of metabolite identification. IMS combines mass spectrometry and spatial information, enabling the visualization and localization of metabolites within tissues or cells. This technique facilitates the understanding of metabolic heterogeneity, biomarker distribution, and disease mechanisms in a spatial context. In targeted Metabolomics Services, a predefined set of metabolites is analysed, focusing on specific pathways or known biomarkers. This approach allows for precise quantification and comparison of metabolite levels between different samples. Targeted metabolomics services offer customized panels tailored to specific research needs, providing accurate measurements for hypothesis-driven studies and biomarker validation. Untargeted metabolomics aims to comprehensively analyse all detectable metabolites in a given sample. It provides an unbiased approach, uncovering novel biomarkers, identifying metabolic signatures associated with diseases or treatments, and generating hypotheses for further investigation. Untargeted metabolomics services employ powerful data analysis tools to mine vast datasets, facilitating the discovery of previously unknown metabolic pathways and interactions. Over the forecast period, the worldwide Western Blotting Market is anticipated to be driven by the use of Western Blotting for medical diagnosis. For instance, the National Centre for Biotechnology Information (NCBI) reports that in April 2021, western blotting was frequently employed in illness diagnosis, mostly for the identification of HIV. Metabolomics Services has revolutionized biomarker discovery by identifying specific metabolite patterns associated with various diseases. Advanced metabolomics services enable the identification of potential biomarkers for early disease diagnosis, disease progression monitoring, and treatment response evaluation. Metabolomics plays a crucial role in understanding drug metabolism, toxicity, and efficacy. Advanced analysis services aid in identifying metabolites generated from drug metabolism, studying drug-drug interactions, and optimizing therapeutic interventions for personalized medicine. Metabolomics Services play an increasingly important role in clinical diagnostics, offering a powerful tool for identifying and monitoring disease biomarkers, monitoring the efficacy of treatments, and supporting personalized medicine. In this article, we will explore the key responsibilities of metabolomics services in clinical diagnostics. Identification and Monitoring of Disease Biomarkers - One of the primary responsibilities of metabolomics services in clinical diagnostics is the identification and monitoring of disease biomarkers. Biomarkers are measurable indicators of a disease state or physiological condition, and they play a critical role in the diagnosis and monitoring of disease progression. Metabolomics Services can analyze the metabolic profiles of biological samples, such as blood, urine, or tissues, to identify the metabolites that are differentially expressed in disease states compared to healthy controls. These differentially expressed metabolites can serve as biomarkers for disease, providing valuable information for diagnosis, prognosis, and monitoring of disease progression. For example, metabolomics services have been used to identify biomarkers for a range of diseases, including cancer, diabetes, and neurological disorders. By analyzing the metabolic profile of biological samples, metabolomics services can identify metabolites that are associated with these diseases, which can be used for early detection and monitoring of disease progression. Monitoring the Efficacy of Treatments - Another important responsibility of metabolomics services in clinical diagnostics is monitoring the efficacy of treatments. Metabolomics services can track changes in the metabolic profile of biological samples before and after treatment, providing valuable information about the effectiveness of different therapies. For example, Metabolomics Services can be used to monitor the efficacy of cancer treatments, such as chemotherapy. By analyzing the metabolic profile of cancer patients before and after treatment, metabolomics services can identify changes in the metabolic pathways involved in cancer growth and proliferation, which can be used to monitor the effectiveness of the treatment. Metabolomics services can also be used to monitor the efficacy of dietary and lifestyle interventions, such as changes in diet or exercise. By analyzing the metabolic profile of individuals before and after these interventions, metabolomics services can identify changes in the metabolic pathways involved in disease progression, providing valuable information for personalized medicine. Supporting Personalized Medicine - Metabolomics Services also play a critical role in supporting personalized medicine. Personalized medicine involves tailoring medical treatment to the individual characteristics of each patient, based on their genetics, environment, and lifestyle. Metabolomics services can analyze the metabolic profile of an individual, identifying biomarkers of disease and response to treatment. This information can be used to personalize treatment plans for individual patients, optimizing treatment efficacy and minimizing side effects. For example, metabolomics services can be used to identify biomarkers of response to chemotherapy in cancer patients. By analyzing the metabolic profile of individual patients, metabolomics services can identify those patients who are likely to respond to a particular chemotherapy regimen, and those who are not. This information can be used to personalize treatment plans for individual patients, optimizing treatment efficacy and minimizing side effects. Quality Control and Standardization- Another important responsibility of metabolomics services in clinical diagnostics is quality control and standardization. Metabolomics services must adhere to strict quality control and standardization procedures to ensure the accuracy and reproducibility of their results. Quality control and standardization procedures can include the use of standardized protocols for sample collection, handling, and analysis, as well as the use of reference materials and quality control samples to ensure accuracy and reproducibility. Metabolomics Services must also ensure the accuracy and reliability of their data through rigorous validation procedures, such as cross-validation and external validation. Metabolomics services play a critical role in clinical diagnostics, offering a powerful tool for identifying and monitoring disease biomarkers, monitoring the efficacy of treatments, supporting personalized medicine, and ensuring quality control and standardization. An Ophthalmoscopes Provides Practitioners With Access To The Retina, Optic Disc, And Other Eye parts13/3/2023 Ophthalmoscopes are essential diagnostic tools used by ophthalmologists, optometrists, and other healthcare professionals to examine the eyes. These instruments allow practitioners to examine the retina, optic disc, and other parts of the eye. Ophthalmoscopes are available in various forms, including handheld, desktop, and portable versions. They are used to detect and diagnose various eye conditions, such as glaucoma, cataracts, and diabetic retinopathy. The history of Ophthalmoscopes dates back to the 19th century, when ophthalmologists first began to understand the anatomy of the eye. In 1851, Hermann von Helmholtz, a German physician, invented the first ophthalmoscope. Helmholtz's instrument used a mirror to reflect light into the eye, allowing the observer to examine the retina. In 1854, Charles Babbage, an English mathematician and inventor, designed a more advanced version of the ophthalmoscope that used a concave mirror to focus light on the retina. The Babbage ophthalmoscope was the first instrument to allow ophthalmologists to view the retina in detail. In 1855, the French ophthalmologist Albrecht von Graefe improved upon Helmholtz's design by adding a convex lens to the ophthalmoscope. This lens allowed for a clearer image of the retina, and Graefe's design became the standard for ophthalmoscopes for many years. In the 20th century, advances in technology led to the development of new types of Ophthalmoscopes, including direct and indirect ophthalmoscopes. These instruments were smaller and more portable than earlier models, making them easier to use in clinical settings. Types of Ophthalmoscopes Direct Ophthalmoscope: A direct ophthalmoscope is a handheld instrument that allows the examiner to view the retina directly through the pupil. The direct ophthalmoscope is equipped with a light source that illuminates the retina, making it easier to see the blood vessels, optic disc, and other structures within the eye. The direct Ophthalmoscopes consists of a light source, a viewing aperture, and a series of lenses that allow the examiner to focus the light on different parts of the retina. The examiner must be very close to the patient's eye to use a direct ophthalmoscope effectively. Indirect Ophthalmoscope: An indirect ophthalmoscope is a larger instrument that provides a wider view of the retina. This type of ophthalmoscope is not held directly against the eye but instead is held a few inches away. The indirect ophthalmoscope consists of a light source, a headband, and a handheld lens. The examiner uses the lens to focus the light on different parts of the retina. The indirect ophthalmoscope is particularly useful for examining the peripheral retina, which cannot be seen with a direct ophthalmoscope. It is also useful for examining the retina of patients with small pupils or cataracts. Portable Ophthalmoscope: A portable ophthalmoscope is a compact version of the direct ophthalmoscope that is designed for use in remote or emergency settings. Portable ophthalmoscopes are battery-operated and often include a rechargeable battery. The portable ophthalmoscope is lightweight and easy to use, making it ideal for use in situations where mobility is necessary. Portable ophthalmoscopes are commonly used in rural areas, where access to healthcare facilities may be limited. Retinal Camera: A retinal camera is a specialized type of ophthalmoscope that captures digital images of the retina. Retinal cameras are used to document and monitor changes in the retina over time, particularly in patients with conditions such as diabetic retinopathy or macular degeneration. Cervical Dysplasia is a disease in which irregular cell growth takes place on the surface layer of the cervix or endocervical duct, the opening amidst the uterus and the vagina. It is also known as cervical intraepithelial neoplasia. It is potentially linked with STDs or HPV contagion, cervical dysplasia is most frequent in women below the age of 30 however can occur at any age. Cervical Dysplasia generally indicates no symptoms, and is most usually exposed by a regular Pap test. The prognosis is exceptional for ladies with cervical dysplasia who get accurate follow-up and therapy. However women who go untreated or who did not receive correct and suitable care are at huge threat of evolving cervical cancer. Normal cervical dysplasia often resolves without therapy, and may only need cautious monitoring with follow up Pap trial. However moderate to serious cervical dysplasia generally needs therapy to reduce the abnormal tissues and decrease the threat of cervical cancer. Often, mild dysplasia that has persevered more than two years may also be cured. In several ladies with Cervical Dysplasia, HPV is seen in cervical tissues. HPV contagion is very general in male and female, and most frequently impacts the sexually active female below 30 years. As per KCC, around 250,000 and one million women entirely the US get affected with cervical dysplasia annually. In many conditions, the immune system reduces HPV and cleans the inflammation. However in few ladies, the contagion endures and causes cervical dysplasia. Above 100 various strains of HPV, above one-third of them can be STDs, and two main kinds are -- HPV 16 and HPV 18 -- are potentially linked with cervical cancer. HPV is generally passed from one person to other person through sexual contact such as vaginal contact, anal interaction, or oral intercourse. However it also can be transferred by any skin-to-skin interaction with a contagious person. Once started, the virus is proficient of increasing from one organ of the body to other, comprising the cervix. Cervical Dysplasia is usually identified over a regular pap test. For this test, the physician swabs the cervix to gather a specimen of tissues. This is usually not aching. The tissues are then sent to a laboratory. It can take around 3 weeks for the laboratory to implement the test. The pap test outcomes can be regular, indecisive, or irregular. If normal, one should survey AAFP’s commendation for proper pap tests. Indecisive results do not show cervical dysplasia. One could have a simple inflammation in the cervix or vagina. The physician may order a recurrent pap test. Furthermore action or analysis will be based on the age and medical background. An irregular outcome is called as cervical dysplasia. It is known as a squamous intraepithelial lesion. On pap test, the precancerous tissues may be categorized as low-grade SIL, showing mild disorder, high-grade SIL, showing mild to serious disorder and ASCUS. Extra testing is required to check if the cell mutations are low, mild, or serious. The term "Liquid Biopsy" describes the examination of any tumor-derived material present in the blood or any other bodily fluid. As the tumour is frequently difficult to access and may need an invasive surgery that could be hazardous, this idea is especially pertinent to lung cancer. Furthermore, the variety of anticancer medications and their sequential application emphasise how crucial it is to carry out an iterative assessment of tumour biology. With the help of liquid biopsies, regional and temporal heterogeneity can be overcome while monitoring treatment response and examining the genetic changes at resistance, as well as noninvasively detecting any targetable genomic alteration and directing matching targeted therapy. Liquid Biopsy has the potential to assist in the management of non-small cell lung cancer at all stages of the disease, including screening, the identification of minimal residual disease to direct adjuvant therapy, early relapse detection, the initiation of systemic treatment and monitoring of response (targeted or immune therapy), and resistance genotyping. The therapy landscape for advanced non-small cell lung cancer has changed as a result of targeted medicines and, more recently, immune checkpoint inhibitors (ICIs) (NSCLC). Using companion biomarkers, response to these drugs can be anticipated. The development of minimally invasive or noninvasive procedures, which yield small tissue samples with extremely small amounts of DNA, creates a dilemma between the necessity to gather large samples for repeated analysis for an increasing number of molecular indicators. In 10–20% of patients, cytological samples from procedures like endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) are insufficient for a thorough molecular analysis. Furthermore, a demand for quick, noninvasive, repeatable assays to evaluate and track tumour biology through therapy has arisen as a result of a better understanding of the resistance to matching targeted therapies. In light of this, there has been a resurgence in interest in Liquid Biopsy, a term used to describe any tumor-derived material that is present in the blood or any other body fluid. The two most extensively researched substrates in the field of NSCLC, circulating tumour cells (CTCs) and circulating tumour DNA (ctDNA), have diverse benefits and drawbacks, and may even work in tandem. Liquid biopsy can noninvasively probe a tumour's molecular landscape (taking into account different clones present within all metastatic sites) and can follow subclonal evolution through iterative blood draws, whereas tissue only provides a snapshot of the tumour at a specific time and location. Whole-exome sequencing offers the capacity to both identify and investigate novel molecular mechanisms of resistance while simultaneously detecting predicted oncogenic drivers or mechanisms of resistance. The majority of clinically significant fusions, however, take place in noncoding areas and are not detected by the whole-exome technique. The use of panels of primers/probes targeting hotspots or exons of specified genes seems to be the most reasonable method in the context of liquid biopsy, given the trade-off between the depth and volume of the genome covered, given the extremely low proportion and amount of cancer DNA. Combined capture NGS is a well-known strategy in the Liquid Biopsy industry. Hybridization to biotinylated probes "captures" predetermined DNA sequences. The leftover DNA can be removed since the biotin is attached to streptavidin beads. These approaches can accurately estimate copy number changes because they avoid using prior amplification. One drawback of hybrid capture for plasma genotyping is the inherent low input DNA, necessitating systems with a high depth of sequencing. Thus, the possibility of sequencing mistakes and false positives exists. For instance, a recent study found that, using tissue as a reference, specificity was 63.5%. For pre-treatment and post-treatment specimens, the concordance rates between cfDNA and tissue were only 64.7% and 48.9%, respectively. RNA interference is monitoring process of many eukaryotic cells that utilize small double-stranded RNA particles as triggers to straight homology-reliable monitoring of gene action. Referred as small RNAs technologies, these ∼21–22 bp long double standard RNA particles have typical 2 nt 3′ overhangs that enable them to be identified by the enzymatic working of RNAi Technology, which further leads to homology-based reduction of the target mRNA. In mammalian tissues, small interfering RNAs are generated from cleavage of huge double stranded RNA antecedents by the RNase III endonuclease Dicer, or they can be manufactured by chemical or biochemical processed. Dicer is combined with RNA-binding agents, the TRBP, PACT, and Ago-2, which are included in the section-off of single interfering RNAs to the RISC. The main compound of RNA-induced silencing complex are the Ago family members. In people there are eight members of this category however only Ago-2 have an active catalytic feature for cleavage action. Whereas single interfering RNAs laden into RNA-induced silencing complex are ds, Ago-2 cuts and discharges the passenger strand, resulting an activated type of RNA-induced silencing complex with a siRNA molecule that shows the particularity of the target identified by intermolecular base pairing. Rules that regulate discernment of strand laden into RNA-induced silencing complex are depending on various thermodynamic abilities of the ends of the single interfering RNAs. The less thermodynamically constant end is ideal for combining to the PIWI domain of Ago-2. RNAi Technology is versatile, and there are several pathways in which dsRNAs control gene expression. The endogenous miRNA pathway starts with Pol II-transcribed main miRNAs that are functioning in the nucleus to pre-miRNAs, transferred to the cytoplasm, and administered again into working miRNAs. The main work of miRNAs is to prevent transformation through partial Watson-Crick base combination to the 3′ untranslated parts of targeted micro RNAi. Otherwise, effortlessly duplexed siRNAs can be generated intracellularly or complete exogenously to tissues. The guide strand is inoculated into the RISC, while it directs sequence-particular reduction of the target transcription, regardless where the base combination takes place. The miRNA and siRNA paths are substitutable, and the essential determinants are the places in the message and the amount of base combination with the targeted transcriptions. Single interfering RNAi can also target transcript gene silencing through connections with chromatin, while they monitor histone and DNA methylation causing inactive chromatin. An essential arm of RNAi includes the microRNAs. These are essential duplexes that posttranscriptionally control gene appearance by completing with RISC and combining to the 3 UTRs of target sequences through short elasticities of homology, termed seed sequences. The main machinery of action of miRNAs is changing repression, though this can be attended by message deprivation. The miRNA duplexes have imperfect Watson-Crick base combination, and the antisense strand can’t be selected by cleavage of the passenger strand as it is for siRNAs; hence the antisense component must be selected by a substitute mechanism. miRNAs are extreme substrates for the miRNAi. The pre-miRNAs are later administered in the cytoplasm by Dicer and one out of two strands is laden into RISC, apparently from interface with one of the Dicer accessory proteins. Essentially, it is likely to exploit this original gene silencing path for regulating genes of choice. Metabolic Testing; Is Used To Measure The Amount Of Calories The Body Burns Throughout The Day27/12/2022 Metabolic Testing includes a number of various tests. Each test offers data regarding a key element of the metabolism. These elements comprises the Resting metabolic rate. Testing this feature of the metabolism offers data regarding the approximated count of calories one burns when the body is at rest as one is not exercising or moving. In maximum capacity of oxygen, the test element (also known as aerobic capacity) offers data regarding the body’s capability to utilize oxygen efficiently over exercise. Lactate threshold test lactates inception in the point at which lactic acid makes up in the plasma more rapidly than it can be eliminated over exercise, leading to muscle fatigue. If one is an athlete who’s interested in enhancing performance, one may find this metabolic test element advantageous. Earlier, Metabolic Testing was done in clinical settings. Presently, this kind of trial has become accessible at many gyms and health clubs. As certification is not needed to read the outcomes of metabolic trials, some consumers feel that testing done at fitness clubs is less appropriate than those a clinical expert conducts. If one is interested in having the metabolism tested, talking to a doctor regarding finding a consistent testing facility in the area is advised to people. The cost may differ as well. Metabolic Testing is generally very costly. It’s up to one to decide where they should get a metabolic test done. Finding out whether the insurance covers it might help one to decide. Metabolic testing is usually done in various parts and almost always comprises a calorimetry trial. This test takes 15 to 30 minutes and is done while one is reclining. The device utilized might differ. Some calorimetry trials utilize a mouthpiece one breathe into. Others need to recline under a plastic hood that’s linked to a monitor by a tube. This test quantifies the RMR. V02 max is resolute while one performs an aerobic activity, like walking or running on a treadmill. One breathes into a mask while this test is being performed. The rising prevalence of lifestyle diseases such as cancer, diabetes and cardiovascular problems is expected to increase the count of people using metabolic testing. A report by NCBI states that around 12 million people in Japan suffer from diabetes. When the metabolic test is completed, one receive the outcomes instantly after and a technique will go through them with individual. When one learns the peak of the persistence and the amount of oxygen one take in, individual can learn the ways to ignore overtaxing oneself and the ways to manage the workouts. If one have been facing problems with weight loss and regulation, are looking for novel methods to push the body, or simply wish to try something innovative for the fitness of the body and complete wellness, catabolic testing could be what one is looking for. With the data metabolic testing returns, one regain a severe benefit and control over the health and wellness. People of all categories can be profited from Metabolic Testing such as athletes, coaches, trainers, and others who just want to keep oneself strong and fit as fiddle can make metabolic testing function for them. |
Categories
All
|