|
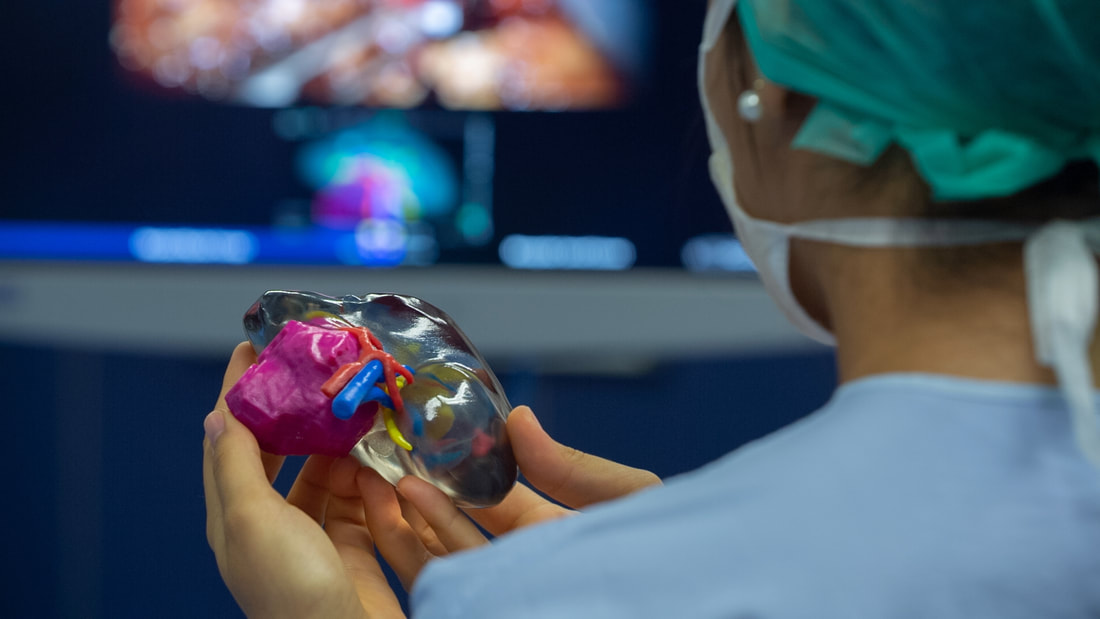
3D Printed Medical Devices are made from plastic, which can present a variety of biocompatibility concerns, as the process involves tight chemical and physical parameters. Plastic devices are more often made from novel materials, which are difficult to analyze. Modeling of patient anatomy is an important step in developing 3D Printed Medical Devices. The process involves digitizing real anatomical structures and segmentation of volumetric images to isolate the structures of interest. The techniques used to segment the images vary depending on the anatomical subject, imaging quality, and scanning modality. Advanced segmentation programs can facilitate this process. Biocompatibility testing is a necessity for medical device manufacturers, especially those with complex design geometries. This process is based on ISO standards and the duration of contact with the device. There are several options available for biocompatibility testing for 3D Printed Medical Devices. For instance, animal tests or living cell tests can be performed to determine whether a device is safe. However, regulators discourage the use of animals for unnecessary testing. As an alternative, a chemical analysis of a device can be used as well. A toxicologist can interpret the data from these analyses. A 3D-printed patient anatomy model can help doctors determine the best way to approach a procedure. This will save both time and money. It is an important part of preoperative planning and procedure rehearsal. 3D-printed anatomic models can also be used to guide surgeons during surgery. The accuracy of a 3D Printed Medical Devices can increase surgical knowledge and improve patient safety. The models can help surgeons plan an operation and reduce the risk of complications. They can also test new techniques before entering the operating room. This helps reduce postoperative stay and reintervention rates. Designing for repair of 3D Printed Medical Devices requires several key steps to make them safe, durable, and reusable. It starts with defining clinical appropriateness, and continues through the development of certification standards and best practices. The standardization process is often complicated and difficult to implement and the advances in the field have made it easier to understand. The process of designing and producing additively manufactured medical devices involves a complex web of links, including a medical-engineer interface, raw materials, printing equipment management, processing parameters, and finished products. In order to ensure quality, the Alliance wants to see the manufacturing of 3D Printed Medical Devices meet the same regulatory, quality, and safety standards as traditional manufactured devices. 3D-printed medical devices may be the next big thing in medicine, there are still many challenges to overcome. Here are some of the major issues involved in the development of these devices. These include the need for biocompatibility testing, modeling patient anatomy, designing for repair or remanufacture, and regulation. Before submitting a medical device to the U.S. Food and Drug Administration (FDA), medical devices must undergo biocompatibility testing. These evaluations are critical for ensuring that medical devices do not cause unexpected adverse effects. The ISO 10993-1 standard provides a guide for conducting biocompatibility tests. Regulators need to set rules and regulations for 3D Printed Medical Devices, and they need to be standardized. This is a necessary step for the future of the medical industry. Regulatory frameworks are important, and they should establish a standard that is based on scientific research. The FDA has issued new guidance on the regulation of 3D Printed Medical Devices. This guidance addresses a variety of issues, from the device and manufacturing process to the testing of products. However, it does not address point-of-care manufacturing, a key aspect of the process.
0 Comments
Leave a Reply. |
Categories
All
|