|
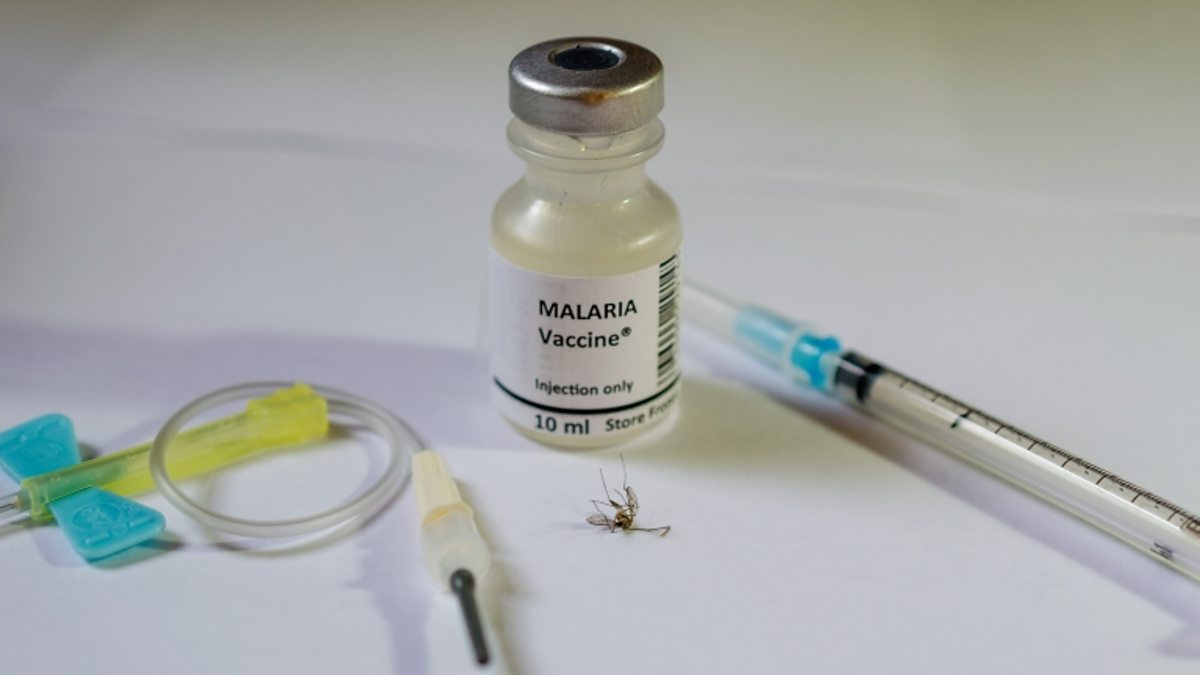
Malaria is a deadly disease that kills over 400,000 people a year, mostly children. Mutating parasites and growing resistance to drugs and insecticides have stalled progress toward a malaria-free world. The World Health Organization has endorsed the first-ever malaria vaccine, RTS, S/AS01. It is the result of 30 years of research and development by GSK, PATH, and partners. A Malaria Vaccines are the medicine that reduces the risk of contracting malaria. It can be administered to people living in endemic areas of the world or traveling there. Several different types of malaria vaccines are currently being developed for malaria. One type, called a circumsporozoite vaccine (CSV), directs immune responses against the major circumsporozoite protein covering the surface of infecting sporozoites. Another type of Malaria Vaccine, called a pre-erythrocytic vaccine, targets the first stage of the parasite’s life cycle by intercepting sporozoites before they get to the liver. A third type of vaccine, called a transmission-blocking vaccine, prevents the formation of new gametes in an infected host. These are the reproductive cells of the parasite that can be ingested by a mosquito and initiate infection. The safety of a malaria vaccine is largely dependent on the nature of the parasite. Many different genetic variants of the Plasmodium falciparum parasite exist in Africa, and so any new vaccine must be effective against a broad range of these. One approach to malaria vaccine development is to target merozoites during the blood phase of their life cycle. This could be done by using antibodies against a merozoite's surface or by blocking its invasion of red blood cells. But a more promising approach is to target the parasite directly through the immune system. This is a more complex task, and many possible strategies can be used to do so. The RTS, S/AS01E malaria vaccine developed by GSK has been tested in a pilot program and has proven safe and effective in infants. It is a feasible product that can be delivered in settings with routine immunization systems and that increases equity in access to prevention. Malaria is one of the world's deadliest diseases: it kills hundreds of millions of people each year, and many of them are children. Many things can be done to combat the disease, but one of the most promising is a vaccine. However, developing a malaria vaccine requires decades of research. The CDC is working on an experimental vaccine, called R21. The Malaria Vaccine is being tested in 450 children living in Burkina Faso, a country that has high seasonal malaria infections. The vaccine's efficacy was 80% in the trial, but it is important to note that this result is based on data from a very small number of people. Although the vaccine is effective, it also has a high dose that can cause a child to have low hemoglobin levels. This can affect the immune system and cause serious health problems. Malaria Vaccines work by stimulating an immune response against a target. In malaria, this immune response may prevent infection by the parasite. It also may help reduce the symptoms of the disease and/or its effects on human health, such as fever, headache, and joint pain.
0 Comments
Leave a Reply. |
Categories
All
|