|
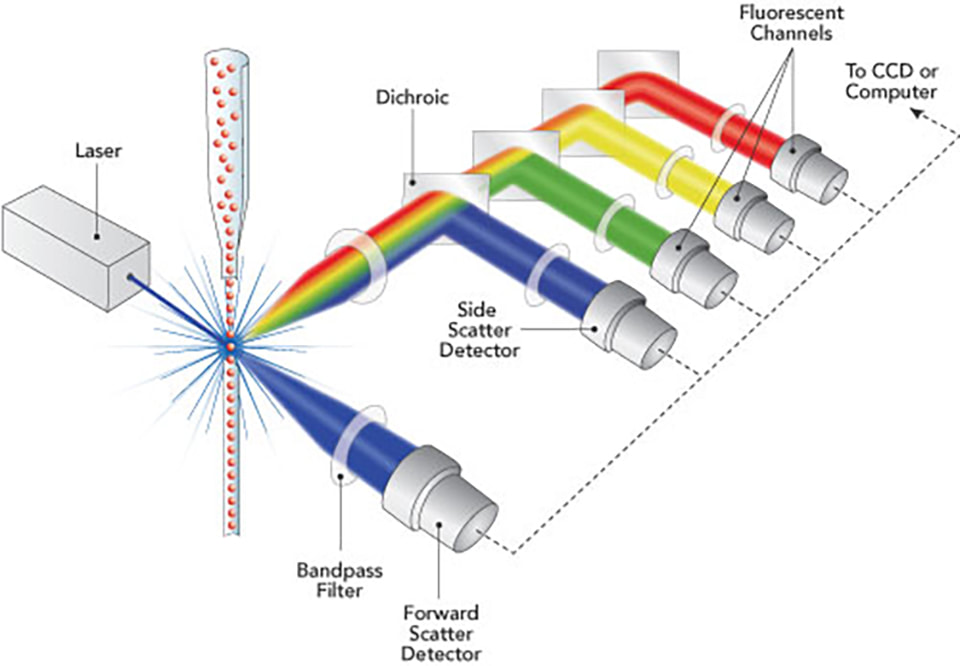
A common cell biology method called Flow Cytometry uses laser-based technology to profile, count, and sort the cells in a heterogeneous fluid mixture. The interaction of cells or other particles suspended in a liquid stream with a laser light beam is monitored by an electronic detecting device as light scatter and fluorescence intensity using a flow cytometer machine. The amount of a cellular component will ideally be represented by the fluorescence intensity if a fluorescent label, or fluorochrome, is precisely and stoichiometrically linked to that component. The automated devices known as Flow Cytometry (FC or FCM) measure the characteristics of one cell at a time. They can quantify a variety of cell properties, including cell size, cell granularity, the quantities of various cell components, including total DNA and recently synthesised DNA, the amount of messenger RNA produced when a gene is expressed, the quantities of particular surface receptors and intracellular proteins, and the quantities of transient signalling events in living cells. When absolute values are required, quantities can be expressed as the number of molecules in a cell. Cell by cell, over 10,000 cells, and up to three to six features or components are often quantitated in a single sample in less than a minute (not counting time to prepare the sample, which might be an hour or more). To distinguish the main groups of leukocytes in peripheral blood, cell size and granularity measurements alone are sufficient. The foundation for clinical equipment that performs automated complete blood counts is this (CBC). Quantitation of particular structures is possible by incorporating fluorescent probes into the cells ("flow cytofluorometry"). The most typical application of Flow Cytometry is for the total DNA per cell in tumour biopsy specimens, for the diagnosis and prognosis of clinical cancer. Quantitation of CD4+ vs. CD8+ T cells in blood is another important application for assessing the effectiveness of anti-HIV medications and determining when an HIV infection has led to AIDS. Research uses Flow Cytometry a lot. In addition to a sizable portion of studies on cell structure, function, and mechanism in other journals, more than one third of papers in the Journal of Immunology also contain flow cytometric data. Monodisperse (single, unclumped) cells are suspended in flow cytometers, where they are passed in single file in front of a laser beam. The amount of dispersed and fluorescent light is measured as each cell moves across the laser beam. Sensitivity is constrained by "autofluorescence," which are naturally fluorescent parts of cells that create a background fluorescence intensity; fluorescent probes must generate much higher intensities for their signals to be reliably quantitated. The majority of Flow Cytometry is analytical; after the data is collected as the material moves through the cytometer, it is discarded. Preparatory flow cytometry sorts living cells into several containers according to their individual characteristics. Fluorescence microscopy (FM) and flow cytofluorometry (FC) can be compared. For a large number of cells, FC can quantify the total levels of a component per cell (typically 10,000, up to 100,000 easily). Normally, FC is unable to determine a component's location within a cell. Flow Cytometry reveals whether a fluorescent component is evenly distributed throughout the cell or concentrated in specific anatomical areas, as well as whether the distribution is time-dependent. The following are the aims of the class's Flow Cytometry experiment:
0 Comments
Leave a Reply. |
Categories
All
|