|
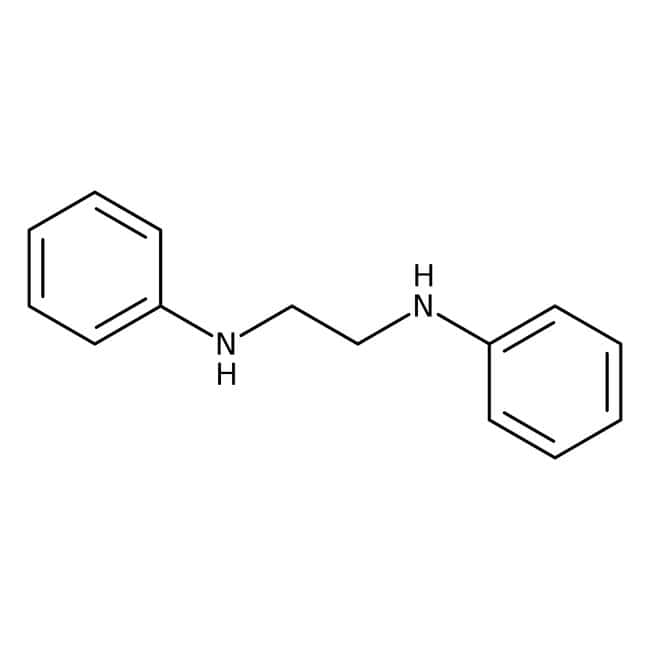
Amines are organic compounds that contain nitrogen atoms with a lone pair. They are derived from ammonia, and thus they are known as arylamines and alkylamines. Amines are organic derivatives of ammonia, in which one, two, or all three of ammonia's hydrogens are replaced by organic groups. Ammonia inorganic derivatives are also known as amines, monochloramine (NClH2), for example. Amines can be found in a wide variety of compounds, including natural and synthetic dyes, vitamins, polymers, and medications. Amine functional groups are also found in many molecules essential to the life, such as amino acids, neurotransmitters, hormones, and DNA. Amines are classified as primary, secondary, or tertiary according to the number of carbons bonded directly to nitrogen atom. Primary amines have one carbon bonded to the nitrogen. While, secondary amines have two carbons and tertiary amines have three carbons bonded to the nitrogen. Amines are named by naming alkyl groups attached to the nitrogen atom, followed by the suffix -amine. In addition to ammonia, amines contain one, two, or all three hydrogen atoms. They also have an aryl and/or alkyl group. Amines are classified based on their general structure and functional groups. Amines with substituents are called aromatic amines. In general, an amine has a boiling point of approximately the same as a hydrogen atom in water. A dilute solution of a strong acid can protone amines, forming salts of alkyl ammonium ions. These compounds can be separated from organic compounds by a simple extraction process. Amines are used to develop chemicals for crop protection, medication, and water purification. They are also used to make azo-dyes and nylon as well as personal care products. U. S. Amines manufactures and markets the world's highest quality synthetic Alkyl & Allyl amines. Ethanol amines are the most common type of amine used in the global market.
0 Comments
Leave a Reply. |
Categories
All
|