|
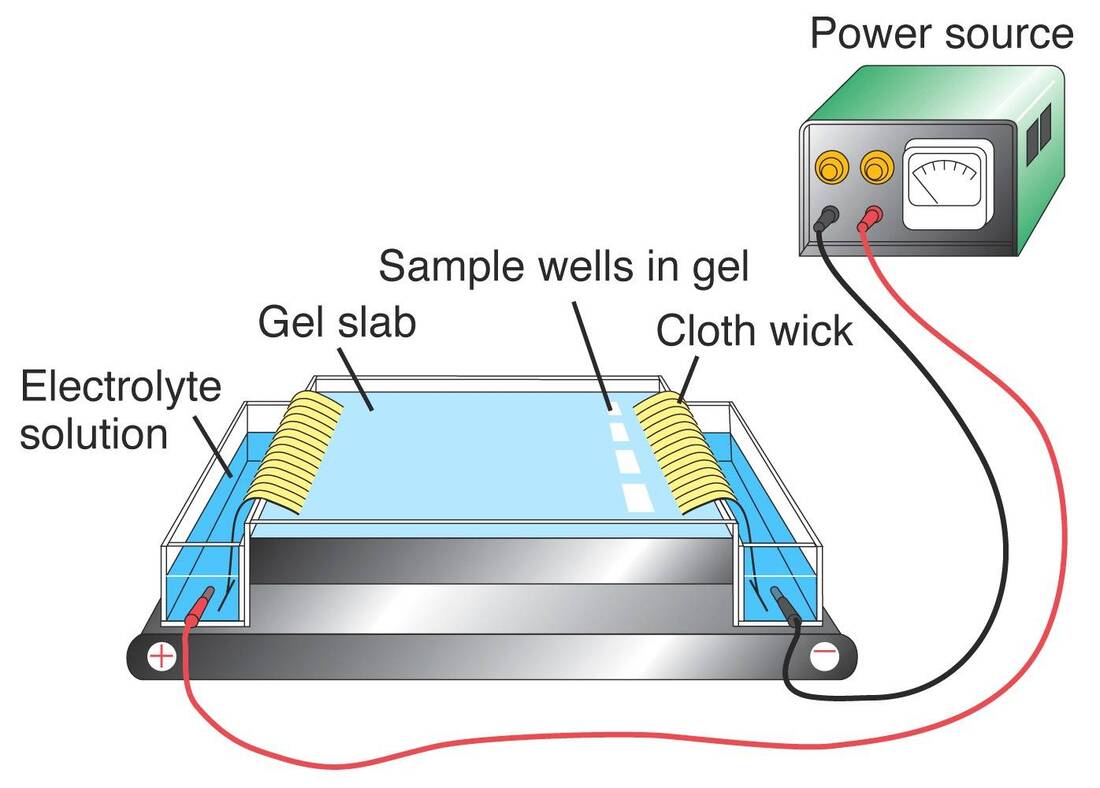
The movement of electrically charged particles in a fluid under the influence of an electric field is known as Electrophoresis, sometimes known as cataphoresis. The phenomena is known as electroosmosis if the liquid itself, rather than the particles, are what are set in motion, such as by a fixed diaphragm. Proteins and other colloids can be analysed and separated using electrophoresis, and it can also be used to cover objects like electron tube components with coatings. The use of electrophoresis as an analytical technique was first developed in 1930 by the Swedish scientist Arne Tiselius. The moving-boundary method of observation was developed by Tiselius, and it involves placing a layer of pure (i.e., particle-free) fluid over a quantity of the same fluid that contains colloidal particles. The boundary between the two layers of fluid is visible and moves at the rate of the particles' Electrophoresis. The mobility of ions in an electric field is the foundation of the separation method known as electrophoresis. Ions that are positively charged move towards a positive electrode whereas those that are negatively charged move towards a negative electrode. One electrode is typically at ground while the other is biassed positively or negatively for safety reasons. Ions can be separated because they migrate at varying rates based on their overall charge, size, and shape. The method is especially useful for macromolecules like proteins. These molecules are given a negative charge, which causes them to gravitate towards the positive electrode. A high-voltage supply, electrodes, a buffer, and a support for the buffer, such as filter paper, cellulose acetate strips, polyacrylamide gel, or a capillary tube, are all components of an electrode device. The other supports are typically employed for biological samples like protein mixes or DNA fragments, while open capillary tubes are used for a wide variety of samples. The support is stained to make it easier to see the separated components after a separation is finished. Isoelectric focusing has a significant positive impact on resolution. The support gel in this method maintains a pH gradient. When a protein descends the gel, it reaches the pH where its isoelectric point is located. The protein concentrates into a distinct band on the gel at this pH because it is neutral and no longer migrates. These are the Specific electrophoretic techniques:
The movement and separation of charged particles (ions) caused by an electric field is referred to as Electrophoresis. Two electrodes (anode, cathode) with opposing charges are joined by a conducting liquid known as an electrolyte to form an electrophoretic system. Differences in the ionic particle's velocity (v), which is the result of the particle's mobility (m) and the field strength (E), cause the separation effect: V= mE A particle's size, shape, charge, and separation temperature all affect an ionic particle's mobility (m), which is constant under specific electrophoretic circumstances. Electrical parameters (current, voltage, and power) as well as characteristics of the medium in which the particles are moving—such as ionic strength, pH level, viscosity, and pore size—are used to describe electrophoretic conditions. One of the main issues with the majority of Electrophoresis methods is the removal of heat produced by the flow of electric current. Every temperature difference alters the speeds of migration through the medium, distorting the bands of molecules that are separated from one another. It is obvious that doing electrophoretic studies at a constant temperature would be optimal.
0 Comments
Leave a Reply. |
Categories
All
|