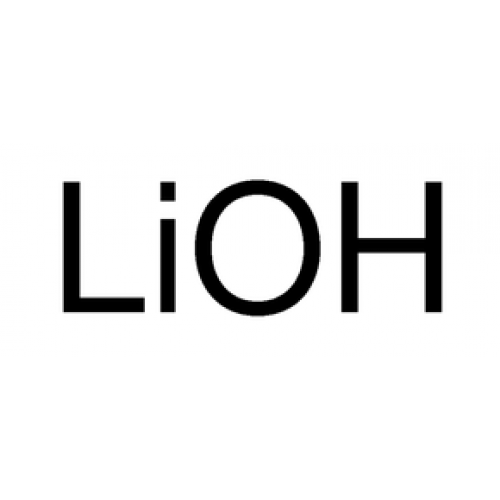|
Lithium Hydroxide is a compound composed of lithium, oxygen, and hydrogen, with the chemical formula LiOH. It is a white, odorless solid that is highly soluble in water, and is commonly used in a variety of applications such as batteries, ceramics, and as a reagent in organic chemistry. Lithium Hydroxide is produced by the reaction of lithium carbonate and water, resulting in the formation of lithium hydroxide monohydrate, which is then dried to form the anhydrous form of the compound. The resulting product is a fine powder that is stable at room temperature and pressure, and has a melting point of 462°C. One of the primary uses of Lithium Hydroxide is in the production of rechargeable lithium-ion batteries, which are used in a variety of electronic devices such as smartphones, laptops, and electric vehicles. Lithium hydroxide is a key component in the cathode of these batteries, which stores the electrical charge that powers the device. Lithium-ion batteries have become increasingly popular due to their high energy density and long cycle life, and lithium hydroxide is a crucial component in their manufacture. In addition to its use in batteries, Lithium Hydroxide is also used in the production of ceramics and glass, where it is added to glazes to improve their properties. It is particularly useful in the production of high-strength ceramics such as those used in aerospace applications, due to its ability to improve the strength and durability of the final product. Lithium hydroxide can also be used as a flux in glass production, where it lowers the melting point of the material and helps to improve its workability. Lithium hydroxide is also used as a reagent in organic chemistry, where it is used to convert carboxylic acids to lithium carboxylates, which can be used in a variety of reactions. It is particularly useful in the production of pharmaceuticals and agrochemicals, where it is used as a key intermediate in the synthesis of a wide range of compounds. In addition to its industrial uses, Lithium Hydroxide has also been investigated for its potential use in the treatment of certain medical conditions. Lithium is a well-known mood stabilizer, and lithium hydroxide has been studied for its potential use in the treatment of conditions such as bipolar disorder and depression. While lithium hydroxide is not used directly as a medication, it is a precursor to the lithium salts used in these applications. Despite its many uses, Lithium Hydroxide is not without its challenges. One of the primary issues is the limited availability of lithium, which is a relatively rare element in the earth's crust. This has led to concerns about the sustainability of lithium mining and production, particularly as demand for lithium-ion batteries continues to grow. In addition, the production of lithium hydroxide can be energy-intensive, requiring significant amounts of electricity to convert lithium carbonate to lithium hydroxide. An inorganic substance, Lithium Carbonate is the lithium salt of carbonate and has the formula Li2CO3. This white salt is frequently used to treat mood disorders and to process metal oxides.
0 Comments
Leave a Reply. |
Categories
All
|