|
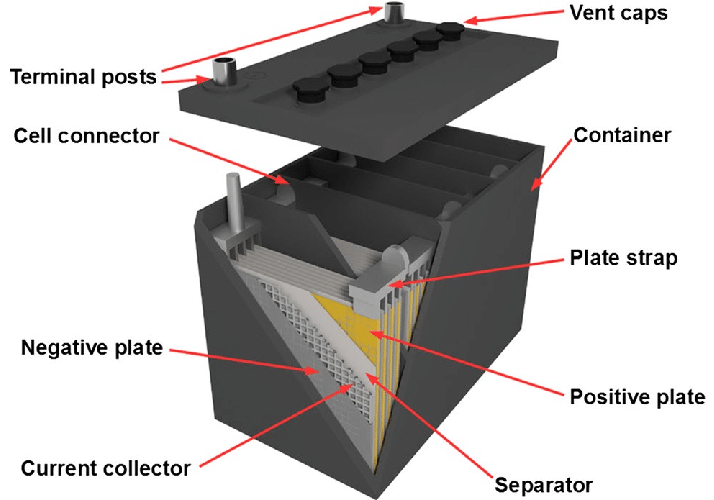
Lead acid batteries are one of the most popular types of batteries. They are used in a variety of applications, including powering electric vehicles and solar panels, as well as uninterruptible power supplies (UPS) and emergency lighting systems. Lead Acid Battery (LAB) is a common energy storage system for off-grid power systems. LABs are made of two electrodes, one a negative cathode and the other a positive anode that are separated by a liquid electrolyte. Sulphuric acid is used as the electrolyte for most lead acid batteries. Lead-acid batteries consist of two electrodes separated by a liquid electrolyte, which is usually a sulfuric acid solution. The negative cathode is composed of sponge lead, while the positive anode is made of lead dioxide. The basic structure of a lead acid battery involves a pair of metal plates made of sponge lead and metallic lead that are immersed in an electrolyte solution consisting of sulfuric acid. During discharge, the plates react with the electrolyte to create water and lead sulfate. During charging, the cells are charged by electron conduction. When the electrodes are removed from the electrolyte, the reaction is reversed. Battery manufacturers are looking to replace the traditional Lead Acid Battery with lithium-ion and nickel-cadmium systems. These lithium and nickel-based batteries are lighter and have better cold-temperature performance, whereas they lose a lot of energy when deep-cycled. Lead acid battery is used in a number of applications including the powering of solar panels. These batteries are ideal for this purpose as they offer high starting currents for a short period of time and are cheap to manufacture. Lead-acid batteries use lead as an anode and lead dioxide as a cathode with an acid electrolyte. These are often considered to be the most effective energy storage materials available, due to their relatively low specific energy and high power density. However, they also have some inherent problems, such as the tendency to corrode and produce toxic vapors. This is why Lead Acid Battery are best avoided in living spaces and never installed in a home without proper ventilation. These batteries can be deep discharged and have a wide range of applications including electric vehicles, large back-up power supplies for telephone and computer centers, off-grid household electric power systems and cellular repeater towers. The major disadvantage of LABs is that they tend to suffer from sulfation in the plate, which reduces their capacity to absorb current. This decreases their longevity. New sealed lead acid (SLA), gel or valve-regulated lead acid (VRLA) batteries have emerged to overcome this problem. A lead acid battery can be used to power a wide range of home appliances, from lights and motors to TVs and refrigerators. It is a versatile power source that’s reliable, inexpensive, and easy to maintain. The Lead Acid Battery consists of three essential components: a sponge metallic lead anode, a lead-dioxide cathode, and an electrolyte made of sulphuric acid. These are connected in a series to produce electricity. It is important to note that lead acid batteries aren’t the most energy efficient power sources. They have limited charge retention and a low cycle life, which means that they can’t be used for long periods of time before requiring maintenance. They also release vapors when charged or discharged that are harmful and can be dangerous. It is crucial to keep them away from children and other people who don’t understand the risks. They should also be installed in a secure place with proper ventilation.
0 Comments
Leave a Reply. |
Categories
All
|