|
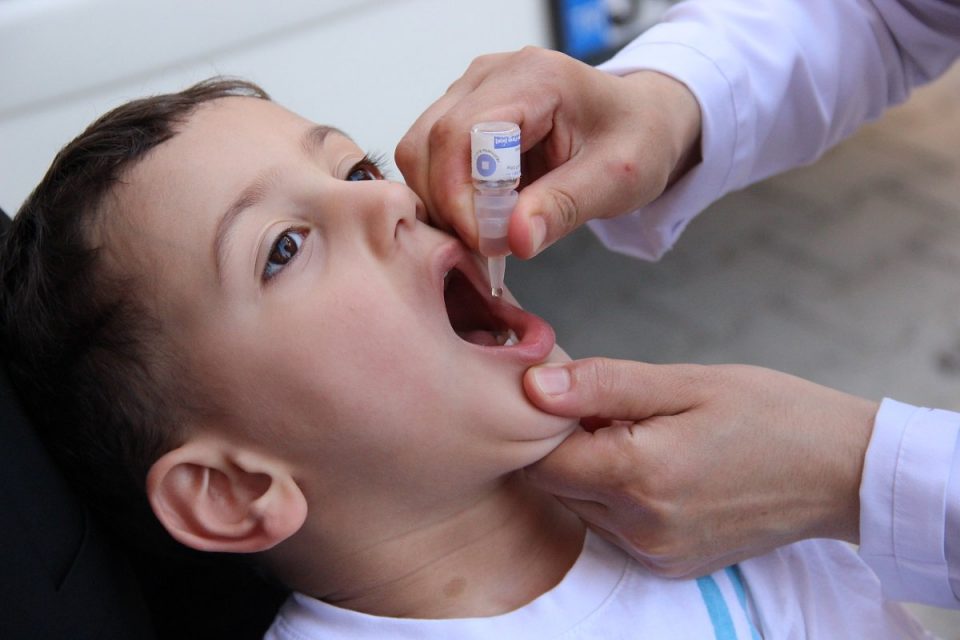
A complete primary vaccination series of three doses are essential to protect against polio. In the USA, 95% of people who receive a full series of Polio Vaccines develop immunity and are protected against paralysis. The most commonly used polio vaccine is trivalent oral poliovirus vaccine (tOPV). It contains live attenuated virus of all three poliovirus serotypes. Vaccination programs are an important part of the polio eradication effort. They are critical for preventing polio outbreaks, stopping the spread of vaccine-derived polioviruses and strengthening routine immunization. To reach high vaccination coverage, governments and international partners need to design and implement effective vaccination programs that deliver multiple doses of oral Polio Vaccines in high-risk countries. This requires detailed planning, meticulous execution, and standardized monitoring and evaluation. These efforts require significant coordination and collaboration across different levels of the healthcare system and society, including the private sector, health workers, local media and religious leaders. Developing these connections can help overcome the barriers to vaccination, particularly among vulnerable populations. Vaccination hesitancy and mistrust of the Polio Vaccines in some communities are often rooted in larger social issues. Using SBC strategies, UNICEF works to increase vaccine acceptance and demand among parents and caregivers in these communities. This can help improve public health messaging, and bolster vaccination coverage. Vaccine research and development is an extensive process, involving many years of laboratory work before a vaccine can be tested in humans. This typically occurs in a university, medical center or small biotech company. During the 1950s, Jonas Salk and Albert Sabin independently developed live-attenuated vaccines against polio. They used animal experiments and cell lines derived from the kidney cells of vervet monkeys to increase the amount of polioviruses in their vaccine. During a global pandemic, vaccine developers, regulators and health authorities have to work quickly to develop, test, license and distribute new vaccines on a scale never before seen. The rapid pace of vaccine development is often accompanied by longer lead times to obtain emergency use authorization and licensure, as well as more complex and costly clinical trial logistics, particularly in low-income countries. Vaccines are complex products, and the manufacturing processes involved can involve a lot of work. These include the use of reagents, testing methods and standards and quality controls that can be time consuming and costly. The Polio Vaccines production process can take up to 36 months, and is subject to many different regulatory and compliance requirements. This includes the need for international standardization of raw materials, the use of ad hoc pharmaceutical quality systems and processes, and a range of tests for safety, efficacy, and regulatory oversight and inspections of the entire manufacturing process. While a number of governments and non-government organizations have been investing in vaccine R&D and production capacity, these resources still do not meet the global need. As a result, countries are often faced with national vaccination policies that are country-dependent and that can pit them against one another in the face of limited supply. This may lead to inequitable distribution, and can increase the risk of outbreaks. Governments have implemented vaccination programs in phases, with populations in each country prioritised according to their risk. This ensures that frontline health workers, the elderly, and others at highest risk of serious disease are vaccinated first. Managing public expectations, however, is crucial to ensure that the process is not viewed as unfair. Vaccine distribution activities should be carried out in a coordinated and systematic manner, using community dialogue and participation as the main driver. Trained locally-based polio workers and volunteers can play an important role in this process, promoting healthy behaviours and building vaccine confidence.
0 Comments
Leave a Reply. |
Categories
All
|